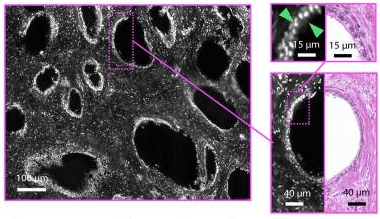
Inverted Light Microscope: A Comprehensive Guide for Students of Microbiology and Laboratory Technicians
A microscope is an essential tool that is used in most laboratories. We would know nothing about the microorganisms around us if this incredible instrument