Need Motor Coordination Mazes for your research?
Introduction
Movement is essential to the survival of every living thing. In order to serve its purpose, this movement must be coordinated, i.e. the nervous system must work to make sure that all movement is directed, regulated and controlled. The loss of this coordination in humans is associated with a range of important diseases, including ataxia, Parkinson’s Disease and motor neuron disease; hence, understanding the neural basis of coordination is crucial in the study of disease and the development of treatments.
In this article we will begin with an overview of the nature and importance of motor coordination. We will then progress to a discussion of each part of the brain that is involved with motor coordination, outlining in turn the specific function that it is believed to serve. Throughout, methods used for the study of the neuroscience of motor coordination in mouse models will also be indicated and explained.
What is Motor Coordination?
Motion is central to the life of all organisms. In order to stay alive and propagate their genes, it is necessary for living things to constantly engage in behaviors that involve movement, such as searching for food, mating, chasing prey, escaping from predators, building dens and so on. Even the most simplistic organisms engage in movement, such as the protozoan paramecium, which moves through fluid by beating tiny hairs called cilia attached to its cell membrane.
It is not enough however for an organism to be able to move. In order for motion to be useful, it must be both controlled and coordinated. If, for example, a monkey wants to grab a fruit from a tree, it is not enough for the monkey to simply shoot out a limb in a random direction and hope that limb magically connects with a fruit. A multifaceted process must occur involving aspects of sensing, actuation, calibration and regulation to ensure the limb grasps its target.
First, sensors (primarily the eyes in this case) must detect the object of interest and situate it in a spatial model relative to the monkey’s body. Then, the monkey’s brain must command its arm and hand to move towards the object in a grasping motion. This movement must be calibrated by the procedural memory of previous movements; the monkey’s brain knows that the arm needs to move in a certain way at a certain speed in this kind of situation. All the muscles must work in concert, to ensure that the arm movement does not unbalance the rest of the body. And finally, the movement must be regulated, that is smoothed out so it is not jerky.
Motor coordination is highly visible in human life under conditions of both positive and negative extremity. On the positive side, when we see a human performing a sequence of complicated and precise movements, such as a dance or a surgical procedure, we can appreciate the amount of calibration and regulation required to perform this sequence with finesse. Equally, when a human being has, for example, a high blood alcohol concentration, we see that they move in a way that is characteristically uncoordinated, failing to properly sense their environment and calibrate, and sometimes even being unable to initiate certain kinds of movement.
The Neuroscience of Motor Coordination
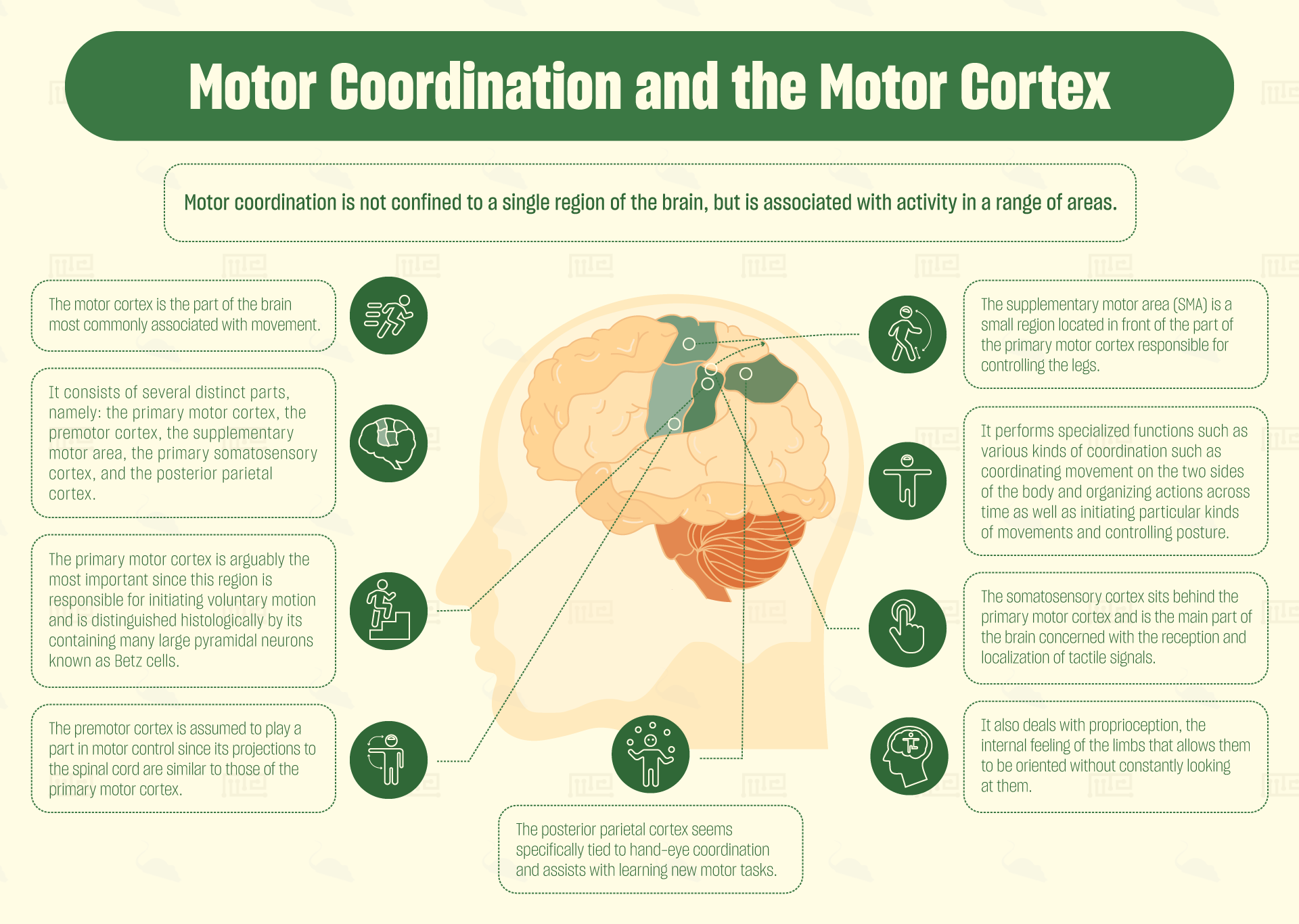
Motor coordination is not confined to a single region of the brain, but is associated with activity in a range of areas. Most commonly highlighted are the motor cortex (which is in fact not one area but a collection of adjacent areas), the cerebellum and the basal ganglia.
The Motor Cortex
The motor cortex, as its name suggests, is the part of the brain most commonly associated with movement. The motor cortex is located predominantly in the anterior part of the frontal lobe and also in some of the parietal lobe. It consists of several distinct parts, namely: the primary motor cortex, the premotor cortex, the supplementary motor area, the primary somatosensory cortex and the posterior parietal cortex.[1]
Of these, the primary motor cortex is arguably the most important, since this region is responsible for initiating voluntary motion. The primary motor cortex is divided into sections, each of which corresponds to a different part of the body (forming a “homunculus”). It controls these body parts by sending signals down cranial nerves to the spinal cord, where they then synapse onto motor neurons. The primary cortex is distinguished histologically by its containing many large pyramidal neurons known as Betz cells.
Research has shown that when an ischemic lesion is administered to the part of the primary motor cortex in mice responsible for controlling their forelimbs, they show a distinct forelimb-dragging behavior in the cylinder test, indicating an inability to control the affected limb.[2] The cylinder test is a simple protocol in which the mouse is placed inside a plastic cylinder and exhibits a natural behavior of attempting to escape by rearing up against the wall. Researchers can then observe the experimental mice and easily see if their ability to rear is deficient compared to controls.
Just in front of the primary motor cortex is a brain region containing a much lower concentration of Betz cells. This has been termed the premotor cortex. This cortex is assumed to play a part in motor control, since its projections to the spinal cord are similar to those of the primary motor cortex. Nonetheless, no-one has so far been able to determine precisely what function the premotor cortex performs in the process of voluntary movement.
Some studies suggest that the premotor cortex assists with more complicated movements, especially those requiring a lot of sensory calibration.[3] While a view seems to have emerged amongst some that the premotor cortex merely plans movements, whereas the primary motor cortex is responsible for actually carrying them out,[4] studies have long shown that both regions are capable of initiating movement and neither is truly subordinate to the other.[5]
The supplementary motor area (SMA) poses even more of a mystery. It is a small region located in front of the part of the primary motor cortex responsible for controlling the legs. In non-human primates, this region contains a body-map, but it does not appear to do so in humans. Specialized functions proposed for the SMA include various kinds of coordination such as coordinating movement on the two sides of the body[6] and organizing actions across time.[7] Other functions proposed include initiating particular kinds of movements as well as controlling posture.[8] It may be that the SMA is a remnant from human ancestors who needed to be capable of dexterously climbing trees.
The somatosensory cortex sits behind the primary motor cortex, and is the main part of the brain concerned with the reception and localization of tactile signals. Like the primary motor cortex, the somatosensory cortex contains a body map, but this map localizes feelings of touch to different parts of the body. This part of the cortex also deals with proprioception, the internal feeling of the limbs that allows them to be oriented without constantly looking at them. Hence, the somatosensory cortex assists in providing feedback and calibrating movements.
The final area of the motor cortex to consider, the posterior parietal cortex, seems specifically tied to hand-eye coordination. Patients with lesions in the posterior parietal cortex find it difficult to focus on and grasp objects.[9] Human PET studies also suggest that the posterior parietal cortex assists with learning new motor tasks.[10]
Cerebellum
The cerebellum, whose name means “little brain” is a walnut-shaped structure that sits below the cortex at the back of the brain, connected to another subcortical region called the pons. The full importance of the cerebellum is still being uncovered, with recent discoveries highlighting its contributions to cognition, but it is most frequently associated with motor coordination.
Humans with lesions in the cerebellum do not suffer any inability to initiate movements, but their movements are no longer well organized in space and time, becoming sudden, excessive and unregulated. It seems like coordinated movement requires a sort of balance of “push and pull” i.e. the motor cortex provides the push, initiating voluntary movements, and then the cerebellum provides the “pull”, reining in the excesses of the movement and smoothing it into something more controlled.
Whereas in humans the cerebellum is quite small compared to the rest of the brain, in many other species—including mice—it’s relative size is much greater, perhaps reflecting the faster and more complicated locomotion required by these species. In mice, the relationship between cerebellar activity and different aspects of locomotion has been studied; one paper found that whereas some of the cells in the cerebellum were highly tuned to the speed of movement, some were tuned to the direction, and others to the rhythm of stepping. The authors suggest that the mouse cerebellum plays a role in adjusting these parameters during motion.[11]
One study has shown that damage to the cerebellum in mice affects their ability to learn in tasks that require strenuous motor activity. Specifically, mice with lesions to different parts of the cerebellum all showed reduced performance compared to controls in the Morris water maze.[12] The Morris water maze is a test whose outcome depends on both navigational memory and motor capability, which requires mice to learn the location of a raised platform in a pool of water and swim to it as quickly as possible. Mice who take longer to find the platform are considered to have performed worse.
Basal Ganglia
The basal ganglia are a collection of brain regions found together, beneath the cortex and just above the pons. The main parts making up the basal ganglia include the striatum, the substantia nigra, the subthalamic nucleus, the globus pallidus and the ventral pallidum. The striatum receives neural signals from the cortex and thalamus and relays them to the rest of the basal ganglia. The basal ganglia then feed information back to the thalamus, mostly via the substantia nigra. Regions in the basal ganglia, most notably the substantia nigra, have been associated with motor control and with disorders of motor coordination.[13]
Lesions of the basal ganglia in humans are correlated with a range of motor deficits, predominantly slowing or lack of movement, but also with loss of control over movement intention.[14] It seems that the main role of the basal ganglia in movement is in suppressing the thalamus and cortex, since overactivity in the basal ganglia is associated with cessation of movement. It is widely accepted that the basal ganglia do not play a role in initiating movements, since lesion patients’ ability to do this is not hindered.
Perhaps the most famous motor disorder associated with the basal ganglia is Parkinson’s Disease (PD). PD patients have jerky, shaky, uncoordinated movements and show a characteristic stooped posture with a shuffling gait. This is thought to be caused by the massive loss of dopaminergic neurons in the substantia nigra, leading to a lack of inhibition of thalamic and cortical circuits, and so, unregulated movements. Treating PD patients with the drug L-dopa restores dopamine concentration in the brain and dampens the shaking.
A number of mouse models have been created to facilitate the study of PD, including the strains Thy1-aSyn and Pitx3. The PD-like symptoms of these model mice can easily be seen by their reduced performance in the rotarod and raised beam tests for motor coordination.[15] In the rotarod test, mice are required to run across a rotating cylinder and maintain their balance. The time that it takes each mouse to fall off, or the maximum speed that each mouse can handle before falling off, is measured and the different mice are compared. In the raised beam test, mice are required to walk across a narrow beam, with the time taken recorded as well as how many times they slip or fall.
Conclusion
Neuroscientific research has revealed how various parts of the brain contribute to the coordination of motor behaviors. These patterns of functional specialization are not only seen in humans but also in other animals including mice. This knowledge of how different brain regions contribute to the control of movement, combined with the development of a range of robust experimental techniques, has allowed for scientific studies with humans and model organisms to contribute to the understanding and treatment of important diseases.
For our apparatuses used in assessing motor function see our Catalepsy bar test, Grip Strength test, the Balance Beam, the Parallel Bars, the Geotaxis Test, the Static Rods Test, the Gait Test, the Parallel Rod test, and the Pole test. See our activity range here.
References
- Teka, W. W., Hamade, K. C., Barnett, W. H., Kim, T., Markin, S. N., Rybak, I. A., & Molkov, Y. I. 2017. From the motor cortex to the movement and back again. PloS one. 12(6), e0179288. doi:10.1371/journal.pone.0179288
- Roome, R. B., & Vanderluit, J. L. 2015. Paw-dragging: a novel, sensitive analysis of the mouse cylinder test. Journal of visualized experiments. (98), e52701. doi:10.3791/52701
- Roland, P.E., Skinhoj, E., Lassen, N.A. and Larsen, B. 1980. Different cortical areas in man in organization of voluntary movements in extrapersonal space. J. Neurophysiol. 43 (1): 137–150.
- Weinrich, M. & Wise, S.P 1982. The premotor cortex of the monkey. J. Neurosci. 2 (9): 1329–1345.
- Fulton, J. 1935. A note on the definition of the “motor” and “premotor” areas. Brain. 58 (2): 311–316.
- Brinkman, C. 1981. Lesions in supplementary motor area interfere with a monkey’s performance of a bimanual coordination task. Neurosci. Lett. 27 (3): 267–270.
- Gaymard, B, Pierrot-Deseilligny, C. and Rivaud, S. 1990. Impairment of sequences of memory-guided saccades after supplementary motor area lesions. Annals of Neurology. 28 (5): 622–626.
- Penfield, W. & Welch, K. 1951. The supplementary motor area of the cerebral cortex: A clinical and experimental study. AMA Arch. Neurol. Psychiat. 66 (3): 289–317.
- Cui, H., & Andersen, R. A. 2011. Different Representations of Potential and Selected Motor Plans by Distinct Parietal Areas. Journal of Neuroscience. 31(49).
- Van Mier, H. I., Perlmutter, J. S., & Petersen, S. E. 2004. Functional Changes in Brain Activity During Acquisition and Practice of Movement Sequences. Motor Control. 8. 500-520.
- Muzzu T, Mitolo S, Gava GP, Schultz SR. 2018. Encoding of locomotion kinematics in the mouse cerebellum. PLoS ONE. 13(9): e0203900.
- Lalonde R, Strazielle C. The effects of cerebellar damage on maze learning in animals. Cerebellum. 2. 4. 300-9.
- Lanciego, J. L., Luquin, N., & Obeso, J. A. 2012. Functional neuroanatomy of the basal ganglia. Cold Spring Harbor perspectives in medicine. 2(12), a009621. doi:10.1101/cshperspect.a009621
- DeLong, M., & Wichmann, T. 2010. Changing views of basal ganglia circuits and circuit disorders. Clinical EEG and neuroscience. 41(2), 61–67. doi:10.1177/155005941004100204
- Fleming, S. M., Ekhator, O. R., & Ghisays, V. 2013. Assessment of sensorimotor function in mouse models of Parkinson’s disease. Journal of visualized experiments. (76), 50303