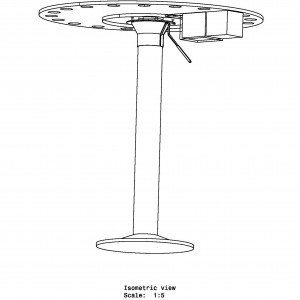
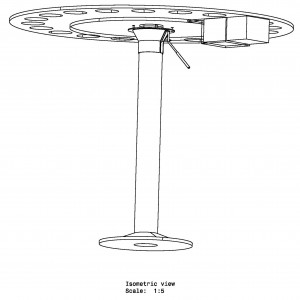
The Barnes maze is a behavioral task used in neuroscience research to assess spatial learning and memory in rodents, particularly mice and rats. Named after its developer, Carol Barnes, the maze consists of a circular platform with multiple evenly spaced holes around its perimeter. One of the holes leads to an escape box or a darkened chamber where the animal can find refuge, while the other holes are dead ends or lead to open spaces without any refuge.
The Barnes maze relies on the natural instinct of rodents to seek safety and escape from open and exposed areas. The task is typically performed in a brightly lit room to create an aversive environment, encouraging the animals to locate the escape box as quickly as possible. It tests the animals’ ability to navigate and remember the location of the escape box based on spatial cues and environmental landmarks.
During the training phase, the animal is initially placed in the center of the maze under a starting cup or covered area. After a predetermined period, the cup is lifted, and the animal is allowed to explore the maze and find the escape box. The animal’s behavior, such as its movement, exploration, and latency to find the escape box, is recorded.
The Barnes maze is often used to assess spatial learning and memory impairments in rodent models of neurological disorders, including Alzheimer’s disease and traumatic brain injury. The task is repeated over several trials, and researchers measure performance parameters such as the time taken to find the escape box, the number of errors made (visiting incorrect holes), and the overall strategy employed by the animal to solve the task.
The Maze Engineers Barnes Maze is available with the following characteristics:
- Includes 1 target box with the option to add more holes
- Easy to clean – removable escape box holder
- Entire top and dark escape box rotate for customization
- False floor modification available for added versatility
- Thick acrylic top eliminates visual cues
- Available in white, grey, clear, or blue
Experience unparalleled versatility and precision with the Barnes Maze – the apparatus of choice for memory and learning research. With its customizable design, easy-to-clean features, and false floor modification, this maze can be tailored to meet your specific research requirements. The thick acrylic top eliminates visual cues in ensuring accurate and reliable results.
Mouse
$ 2290
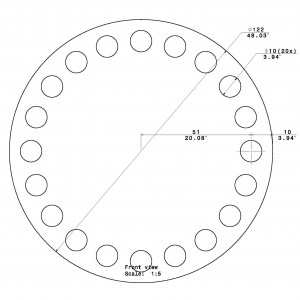
+S&H- 92cm diameter,
- 20 holes,
- 5cm hole diameter,
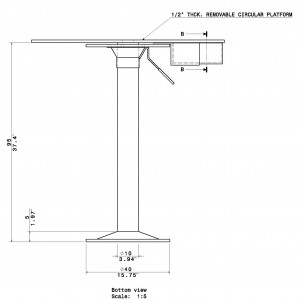
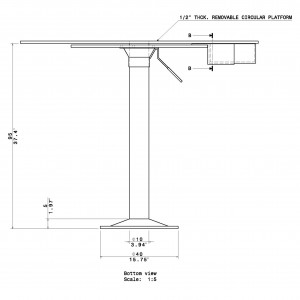
- 95cm stand height (adjustable)
- Removable Top.
- Black Nesting Chamber w clear nest holder included: 20cm x 11.5cm x 4cm (L x W x H)
- Matted finish
Rat
$ 2490
+S&H- 122cm diameter
- 20 holes
- 10cm hole diameter
- 95cm stand height (adjustable)
- Removable Top
- Black Nesting Chamber w clear nest holder included: 22.5cm x 14cm x 9cm (L x W x H)
- Matted finish


Barnes Maze False Floor Add on Mouse $650.00
Barnes Maze False Floor Add on Rat $850.00



Overview
Barnes Maze (BM) is a behavioral task often used in neuroscience for the study of spatial learning and memory. The task’s primary ability is to measure the capacity of the subject to learn the location of the target by using distal visual cues. The maze exploits the averseness that rodents feel towards open and brightly lit spaces to motivate them to find the target location. The Barnes Maze requires the use of hippocampal-dependent spatial reference memory to be able to locate the escape locations. This ability to remember the location of the target hole can be affected by the administration of certain drugs or disease models.
The Barnes Maze was designed by Carol Barnes in 1979 to evaluate spatial learning and memory. Initially intended for rats, the Barnes Maze has been increasingly adapted to be used with mice as well (Sunyer et al., 2007). The BM task draws similarities to the Morris Water Maze (MWM) and the Radial Arm Maze (RAM) task; however, unlike the aforementioned mazes, the Barnes Maze does not expose the subjects to strong aversive stimuli such as forced swimming and food/water deprivation and in comparison can be considered to be a low-stress alternative to these tasks (Harrison et al., 2006). The maze, since its conception, has been used not just for spatial learning and memory but also in testing and validating the effects of drugs and pharmacological compounds in models of diseases like Alzheimer’s (Harrison et al., 2006, Attar et al., 2013), and in understanding learning and memory deficits associated with mild traumatic brain injuries.
The Barnes Maze apparatus is a simple circular platform with circular holes serially placed along the edge of the platform. The task of the subject is to find the hole that serves as the target location. The target location leads to a small and dark recessed chamber beneath the platform and is not visible to the subject from the platform. Intra- and extra-maze cues are often used to assist the subject in finding the target hole.
Variants of the Barnes Maze include the Delayed Matching to Place Barnes Maze, Randomized Barnes, and Radial Arm Barnes Maze.
History
2.1 Origin
The BM was developed by Carol Barnes and described in her paper investigating memory deficits associated with senescence (Barnes 1979).
The effects of 3,4-diaminopyridine in the age-related improvement of short-term spatial memory was investigated using the BM, the results of which suggested that the 3,4-DAP selectively improved memory performance of the old subjects, and, within that age group, only improved performance on the short-term memory task (Barnes et al.,1989).
Markowska et al., 1989 utilized the BM for spatial memory and reversal tasks in their investigation to determine the correlations among different behavioral and neurobiological measures in aged rats.
Since its initial use, the BM has seen a slow but steady growth in use over the years in investigations related to neurodegenerative diseases such as Alzheimer’s and in understanding the effects of brain lesions.
2.2 Developments
Vorhees (1997) in his paper investigated the effects of prenatal exposure to neurotoxins. For his investigation, he used different behavioral assays to assess the different types of learning and memory, one of these tests being the BM task to assess spatial learning. These tasks were used to detect long-term CNS dysfunction after prenatal exposure.
Adult Lhx5-deficient mice were used in the investigation of learning impairments and motor dysfunctions by Paylor et al.,2001. The hippocampus plays a crucial role in memory and learning, and its absence or disorganized neuroanatomy as observed in the Lhx5 mutated mice reflects poor performance in the BM spatial learning task.
The potential of voluntary running in aiding cognitive brain and cognitive functions after Whole-brain irradiation was assessed by Wong-Goodrich et al.,2010. When the subjects were assessed in BM task after daily running following WBI, it was observed that running significantly prevented spatial memory retention decline observed months after irradiation. It was concluded based on their observations that exercise assisted in the recovery of hippocampal plasticity and could be used as a potential therapeutic intervention.
2.3 Recent Developments
Meyer et al.,2014 tested their hypotheses that neonatal leptin would prevent the development of Growth Restricted (GR) associated behavioral abnormalities. In the BM task, the baseline escape times were faster for GR mice; however, the GR mice exhibited regression in their escape times on days 2 and 3. They concluded that alternation in social interactions, learning and activity of mice due to GR could be mitigated by supplementation with the neurotrophic hormone leptin.
The risks of space radiation to astronauts and Alzheimer’s disease-related pathology were evaluated by Rudobeck et al.,2017. APP/PSEN1 transgenic mice and wild-type mice were irradiated with protons, and their performance was tested on the BM at 3 and 6 months after irradiation to evaluate spatial learning and memory.
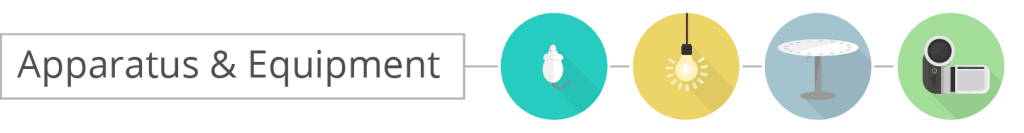
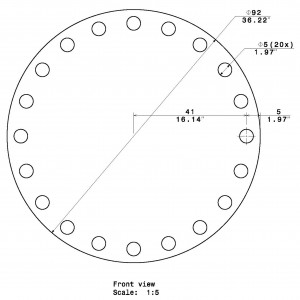
The Barnes Maze apparatus is composed of a circular platform that is raised above the floor to a height of approximately 100 cm. Holes of sizes ranging from 50 to 100 cm in diameter to accommodate mice, rats, and small primates are arranged serially and are equally spaced along the perimeter of the platform.
The holes are placed several centimeters from the edge of the maze, and the number of holes can vary from anywhere between 18 to 20 or more. The designated target hole has a recessed small, dark chamber under the platform. This hole serves as a safe space for the subject and is not visible from the platform to the subject. The entire platform is rotatable around the center point allowing the location of the escape chamber to be easily varied.
The platform of the maze is colored in contrast to the color of the animals being tested. For ease of observation, the holes of the maze are numbered starting from the target hole; the holes to the right of the target hole are numbered starting with +1, and those to the left are numbered starting with -1, with an opposite hole directly across from the target hole, labeled zero.
The visual cues used for the task can be either intra- or extra-maze cues and can be prepared using different colors and shapes. The cues serve as a reference point for the subject during the task.
To avoid shadows in the maze, the BM should be well-lit from above. Proper lighting also ensures that the subject can see the rewards or other cues. Tracking software and video camera, such as Noldus Ethovision XT or ANY-Maze, mounted above the maze can assist with live scoring and tracking, and recording the subject and its movements within the maze. The apparatus must be cleaned thoroughly before and after each trial to limit the influence of any residual stimuli from the previous trials.
Protocol
The Barnes Maze is a simple task used in measuring spatial learning and memory in rodents and small primates. The task measures these parameters by observing the ability of the subject to remember the location of the target hole leading to an enclosed escape chamber.
This test can provide information regarding hippocampal-dependent learning, specifically spatial memory. The BM task has been utilized to understand the effects of age and neurodegenerative diseases on the learning and memory capabilities of the subject. Typically, animals are capable of learning and remembering the location of the target hole using intra- and extra-maze cues.
Before every trial, the apparatus must be thoroughly cleaned to avoid the influence of residual stimuli, if any, from influencing the performance of the subjects.
Pre-Training for the Barnes Maze
The apparatus is set up, and the visual cues are placed in their respective locations. The cues remain constant throughout all training and testing trials. The subject is (usually) placed in a cylindrical dark start chamber in the middle of the circular platform and released after 10 seconds have elapsed.
The subject is gently guided towards the escape hole avoiding any force to prevent unnecessary stress on the animal. The subject is allowed to remain in the escape chamber for 2 minutes.
Evaluation of Spatial Memory Using the Barnes Maze
The apparatus is cleaned to remove residual olfactory cues from previous runs, and the platform is rotated on its central axis to control any remaining olfactory cues. The escape chamber is adjusted so that it is in the same position.
The video recording is started, and the subject is released from a cylindrical chamber from the center of the platform after 10 seconds. The trial lasts about 3 minutes, during which the subject freely explores the platform. Errors are recorded every time the subject pokes its head into a hole that is not the target hole, and the latency time is determined as the time the subject takes to reach the target hole. The trial ends when the subject has entered the escape chamber, or the 3 minutes have elapsed. In the event, the subject fails to find the escape chamber it is gently guided to it and allowed to remain in it for 1 minute. The subject is returned to its home cage until the next trial.
Each animal should perform four trials on each of the four testing days with approximately fifteen minutes inter-trial intervals.
Evaluation of Reference Memory Using the Barnes Maze
On the fifth testing day, the target hole is closed, or the escape chamber is removed. The trial is initiated as mentioned earlier and lasts for 90 seconds. The number of errors and the latency time are recorded. The subject is removed from the maze when the 90 seconds have elapsed.
The procedure is repeated 7 days later, on the twelfth day.
Since its original design, the Barnes Maze has been adapted with several simple modifications. For example, a curtain surrounding the maze platform can be used to prevent the animals from making spatial associations between distal room cues and the location of the target hole (Harrison et al., 2006, Rosenfeld & Ferguson 2014). Protocol variations have also been made to increase task difficulty (Attar et al., 2013).
The escape chamber beneath the maze can optionally lead to an escape tube that allows the animal to reach a home cage or other safe space (Rosenfeld & Ferguson, 2014). A false floor can also be added beneath the maze platform to close off the holes that do not lead to the escape chamber.
The hole positioning has also seen modifications over the years to improve the spatial learning and memory measure of the Barnes Maze task. The Delayed Matching to Place Barnes Maze (DMP Barnes Maze), is a dry variant of the DMP water maze by Steele and Morris (Steele and Morris 1999) that was refined by Faizi et al. 2012. The DMP maze is a Patterned Barnes Maze (PBM) that has the escape platform frequently changed during trials. The apparatus includes an elevated circular platform having 40 holes arranged across the inner, middle and outer rings. Each of these holes is attached to an ABS tube of which only one tube acts as an escape tube.
Another variation of the Barnes Maze is the Randomized Barnes Maze which was designed to overcome the limitation of the BM. In Barnes Maze, the holes are arranged serially along the circumference of the platform that can be serially searched by the subject rather than using a spatial strategy. The Randomized Barnes Maze is modified such that the holes are placed in a pseudorandom order to discourage non-spatial strategies.
A combination of the classic Radial Arm Maze and Barnes Maze, the Radial Arm Barnes Maze combines the advantages of both mazes into one. The maze was first described by Paganelli’s et al. in their 2004 paper investigating the influence of ischemic brain damage on the acquisition and retention of cognition in mice.
Sample Data
The data obtained from the Barnes Maze generally consists of two main measures: the number of error head pokes the animal makes, and the time it takes the animal to enter the target hole and the escape chamber. Other measures such as the total path length and movement speed can also be measured and obtained from video tracking software.
As the animal learns the relationship between local or distal spatial cues and the location of the target hole, the number of error head pokes and the latency time should decrease. These measures can be simply graphed and compared across a sham control group and a disease model.
The search strategy used by the animal must be analyzed manually using the video recording and tracking software. Generally, one of three search strategies is used by the animal to locate the target hole:
- Direct: The animal moves directly to the target hole or an immediately adjacent hole before entering the target hole
- Mixed: The animal searches random holes, crossing through the center of the maze
- Serial: The animal visits several adjacent holes in a clockwise or counter-clockwise manner before reaching the target hole
The exact position of each head poke error can also be counted and graphed to help visualize these strategies.
Using graphs to compare latency time, the number of error head pokes, the position of these errors and the total path length between different disease or treatment groups the effect on spatial memory and learning can be easily visualized.
Animals in the control groups should show significant improvements in reaching the target hole quickly and efficiently. Animals as disease models of neurodegenerative disorders, for example, should show a much slower learning curve with more errors and longer path lengths, even after several days. Generally, animal cohorts of 10-30 animals are sufficient to obtain p-values of <0.05 using ANOVA and step-down Bonferroni tests (Harrison et al., 2006, Attar et al., 2013, Sunyer et al., 2007).
8. Translational Research
The Barnes Maze is a simple and straightforward task to assess spatial learning and memory in neurocognitive diseases, neurodegenerative diseases, and traumatic brain injury models.
The benefits of exercise have often been explored as a therapeutic intervention for cognitive improvement. In their study regarding the effects of Whole-brain irradiation (WBI) therapy, Wong-Goodrich et al. were able to show that running can abrogate the progressive learning and memory deficits induced by WBI and aid in the recovery of adult hippocampal plasticity. In the study that was conducted by Wu et al., swimming exercise was observed as a promising therapeutic option in the prevention of neurodegeneration in the elderly and/or AD population.
Space radiations present a health risk to astronauts spending long missions in space. Rudobeck’s et al. investigation aimed to understand the impact of protons, the main constituent of the space radiation spectrum, in accelerating the onset of Alzheimer’s disease and AD-related pathology.
In comparison to the Morris Water Maze and the Radial Arm Maze, the Barnes Maze is relatively less stressful. The MWM subjects the animal to significantly more stress as the subject must be submerged in water and swim in order to survive and search for the escape platform (Hodges 1996, Harrison et al., 2006). However, some groups report that there is little difference in stress and anxiety between the two mazes (Harrison et al., 2006).
Although the BM is considered a less anxiogenic alternative to other behavioral assays, aversive stimuli such as bright light and adverse noise can be used to encourage explorative drive in finding the escape hole. The absence of significant stressors, such as forced swimming and food/water deprivation, allows for better observations of working and reference memory in the animals as they perform in the maze.
In the absence of aversive stimuli to motivate the subject to seek the enclosed target chamber, the subject may simply explore the maze rather than completing the task. Further, if the maze is being used for multiple animals, proper cleaning of the apparatus is a must to ensure no olfactory cues from previous trials influence the performance of subsequent subjects. This can be easily achieved by cleaning the maze before and after each trial.
As with all mazes that measure aspects of learning and memory, it is important to remember that many different processes play into the behavior observed in the maze. Factors such as anxiety and exploratory activity should be considered when interpreting the results of a spatial memory task.
- The Barnes Maze was designed and developed by Carol Barnes in 1979.
- The Barnes Maze has similar principles as the MWM and RAM; however, unlike those behavioral assays, the BM does not use strong aversive stimulus.
- The Barnes Maze is considered as a less stressful alternative to the MWM.
- The Barnes Maze uses an elevated circular platform that has equally spaced holes placed at the perimeter of the platform.
- The subject’s natural tendency to seek sheltered, dark spaces is challenged by the open and brightly lit platform, forcing it to explore the maze for the target hole.
- Intra- and extra-maze cues are often used to assist the subject in remembering the location of the target hole.
- The difficulty of the maze can be varied by removing the maze cues.
- Animals in control groups show rapid learning as they remember the location of the target hole, while in comparison, animals as disease models of neurodegenerative disorders show a much slower learning curve.
Mouse
Mouse Size (CM)
- Diameter: 92
- 20 Holes
- Hole Diameter: 5
- Stand Height: 95
Rat
Rat Barnes Maze Size (CM)
- Diameter: 122
- 20 Holes
- Hole Diameter: 10
- Stand Height: 95