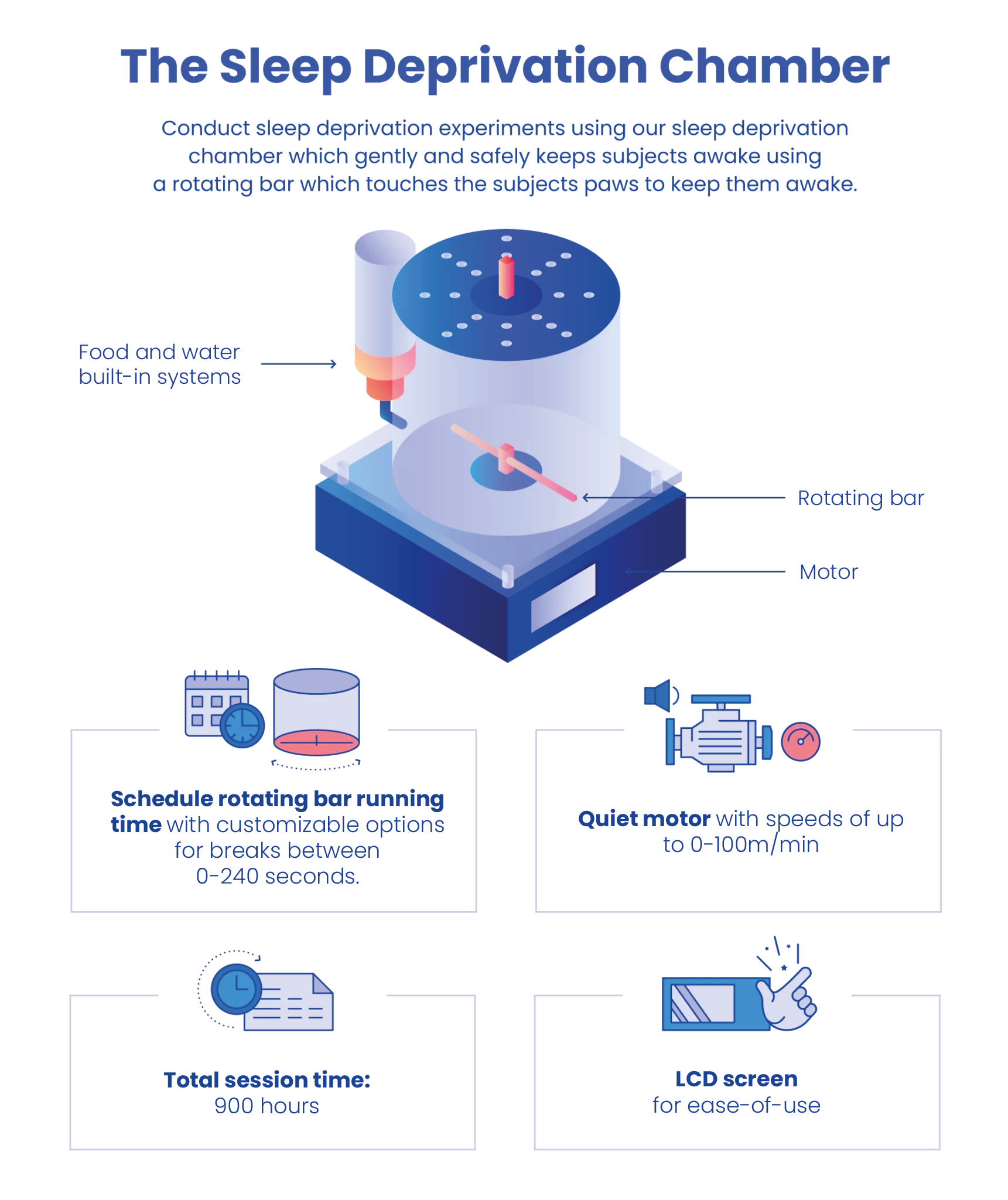
Sleep deprivation (SD) impairs spatial, emotional, and working memories, and augments anxiety-like behaviors. The sleep deprivation chamber model of rodent sleep deprivation apparatus from MazeEngineers is an automated device that provides movement at researcher programmed intervals to prevent REM sleep. The apparatus consists of a clear acrylic chamber atop a motor and screen to set time and rotations of a bar inside the chamber to prevent the subject from sleeping in a safe way. Food and water apparatuses are available to allow long-term experimentation. Lid slots allow for cable and cannula use.
Features
Price & Dimensions
Sleep Deprivation Chamber
$ 4490
Per Month- Chamber dimensions: (D) 28 cm X (H) 25 cm
- Time and rotation apparatus (L) 25 X (H) 25 X (W) 10cm
- Lid slots allow for cables, cannulas for applications in optogenetics, EEG etc
Documentation
Introduction
The Sleep Deprivation Chamber is an automated chamber that can be used for sleep deprivation and sleep fragmentation studies in rodents. The apparatus consists of a cylindrical cage with a rotating bar at its base. The bar stimulates gentling handling, which constantly disrupts rodents’ sleep by touching their feet. It can be set to the desired speed and direction of rotation using the software. Moreover, a random rotation can also be selected to prevent sleep acclimation and habituation. Since the chamber is automated, it minimizes the need for experimenter intervention.
Sleep is essential for the normal functioning of humans. The lack of sleep causes cognitive deficits in waking cognitive functions, observed as the inability to focus, think with clarity, and sustain attention (Worley, 2018). Sleep deficits also have adverse outcomes in children, affecting their cognitive, physical, and socioemotional development (Schlieber & Han, 2021). Therefore, sleep deprivation and sleep fragmentation studies can examine how sleep deficits affect different domains in human cognition and behavior. The Sleep Deprivation Chamber can easily be used to study the effects of sleep deprivation on cognition and behavior using a rodent animal model. It can be used in conjunction with other behavioral testing apparatuses such as the Y-Maze and the Morris Water Maze.
The Sleep Deprivation Chamber also includes built-in food and water systems for long-term experimentation. The chamber is large enough to accommodate multiple subjects to prevent social isolation stress and disrupt the sleep of several subjects simultaneously.
Apparatus and Equipment
The Sleep Deprivation Chamber consists of a cylindrical plexiglass cage. A rotating bar is present at the base of the cage that can be controlled by software to allow the desired speed and direction of the rotation. A moderate rotation speed of 10-40 rotations per minute can be selected to disturb the subjects’ sleep as the bar touches their feet. An optional lid slot allows for the use of cannulas and cables for applications such as Optogenetics and EEG. Video tracking and scoring are available via Noldus EthoVision and ANY-Maze with camera mounts and gantries upon request.
Training Protocol
Clean the cage and change the bedding after every trial. Refill the food and water containers when they are emptied.
Sleep Deprivation Task
Place the subjects in pairs in the Sleep Deprivation Chamber to eliminate social isolation stress. Subject the rats to sleep deprivation for 6 hours a day for seven days. Increase the sleep deprivation duration to 8 hours per day after the seven days. Conduct trials for seven days with the sleep deprivation increment.
The increment in sleep deprivation is necessary for developing rats since the percentage of sleep during the light phase increases with age.
Literature Review
Evaluation of the effect of early life sleep deprivation in rats
Atrooz, Liu, Kochi, and Salim (2019) utilized the Sleep Deprivation Chamber to evaluate the effect of sleep deprivation in the early life of rats. Male Sprague Dawley rats were deprived of 6-8 hours of sleep per day for 14 days beginning at postnatal day (PND) 19. Behavioral tests were conducted at the end of the sleep deprivation task at PND 33. The Open Field Arena was used to investigate exploratory behaviors. Anxiety-like behaviors were assessed using the Light/Dark Box, the Elevated Plus Maze, and the Social Interaction Test. The Forced Swim Test was used to observe depression-like behaviors. The behavioral tests were repeated at PND 60 and 90. Anxiety-like behaviors were observed in the sleep-deprived rats at PND33 and PND60 compared to the control group. However, the subjects showed depression-like behaviors only at PND90 and not during early life. A separate set of subjects were euthanized after the sleep deprivation task for blood collection and brain dissection. Oxidative stress and inflammatory markers were evaluated. Results indicated a significant increase in oxidative stress at PND33 but not at PND90.
Evaluation of sleep deprivation in mice using the Sleep Deprivation Chamber
Naylor, Harmon, Gabbert, and Johnson (2010) evaluated whether gentling handling by the Sleep Deprivation Chamber effectively induces sleep deprivation in mice. Ten wild-type C57Bl6 mice between 11-13 weeks old were used in the study. The mice were implanted with EEG recording electrodes and EMG recording electrodes to measure sleep. The mice’s sleep was then recorded for 24 hours, followed by 24 hours of sleep deprivation and a 12 hours recovery period. The direction of the rotation bar was randomly reversed every 30-90 seconds to prevent acclimation. A yoked control mouse that was independently housed also received 24 hours of sleep deprivation but was allowed to sleep at all other times. Results indicated that the yoked control averaged three times more sleep than the sleep-deprived mice during the sleep deprivation task. The sleep-deprived group had significantly more NREM sleep than the yoked control during the recovery period.
Strengths
The Sleep Deprivation Chamber disrupts rodents’ sleep by gentling handling. The rotating bar gently touches the rodent’s feet for constant sleep disruption. The speed and rotation of the bar can be set according to experimental needs. The subjects are prevented from habituating to the bar rotation by selecting a random rotation. The chamber is automated to minimize experimenter intervention and resources compared to manual deprivation.
Summary
- The Sleep Deprivation Chamber can be used for sleep deprivation studies in rodents.
- It is an automated chamber consisting of a cylindrical cage with a rotating bar at its base. The bar constantly rotates and touches the rodent’s feet to disrupt sleep. The speed and rotation of the bar can be set according to experimental needs.
- The chamber includes food and water apparatuses to allow long-term experimentation.
- The chamber can be used in conjunction with other behavioral and cognitive tests to examine the effect of sleep deprivation on behavior and cognition.
References
- Atrooz, F., Liu, H., Kochi, C., & Salim, S. (2019). Early Life Sleep Deprivation: Role of Oxido-Inflammatory Processes. Neuroscience, 406, 22–37. https://doi.org/10.1016/j.neuroscience.2019.02.021
- Naylor, E., Harmon, H., Gabbert, S., & Johnson, D. (2010). Automated sleep deprivation: simulated gentle handling using a yoked control. Sleep, 12(1), 5-12.
- Schlieber, M., & Han, J. (2021). The Role of Sleep in Young Children’s Development: A Review. The Journal of genetic psychology, 182(4), 205–217. https://doi.org/10.1080/00221325.2021.1908218
- Worley S. L. (2018). The Extraordinary Importance of Sleep: The Detrimental Effects of Inadequate Sleep on Health and Public Safety Drive an Explosion of Sleep Research. P & T : a peer-reviewed journal for formulary management, 43(12), 758–763.
Request a quote
"*" indicates required fields