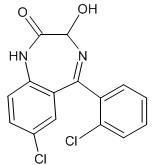
What is Ativan?
Ativan (Lorazepam) is a synthetic drug belonging to the drug class Benzodiazepines and is mainly used to treat symptoms related to anxiety disorders. [1]
Ativan’s appearance is described as a nearly white powder that is virtually insoluble in water.[2]
History of Ativan
Austrian chemist, Dr. Leo Sternbach, accidentally discovered Benzodiazepines in 1954. The drug was discovered to be effective, and by 1959, Chlordiazepoxide (Librium) was introduced to the market.[3]
Ativan followed suit a couple of years later, being developed by DJ Richards and marketed in 1977.[4]
Chemical Structure and Pharmacology of Ativan
Benzodiazepines are made up of a diazepine and a benzene ring, with different side groups attached.[3] The particular side chain determines the nature of its effects.[5] Ativan’s chemical name is 7-chloro-5-(o-chlorophenyl)-1,3-dihydro-3-hydroxy-2H-1,4-benzodiazepin-2-one (C15H10Cl2N2O2) and it has a molecular weight of 321.16 g/mol.[6]
Ativan is absorbed in a very high rate and results in 90% bioavailability when administered orally.[4] Ativan’s volume of distribution is 1.3 L/kg, with 85% of each dosage being protein-bound. Ativan does not redistribute as quickly in the brain due to its lipophilic nature, this contributes to its longer-acting sedative effect.[4],[7]
Metabolism occurs in the liver by CYP450 isoenzymes, which conjugate the drug at its 3-OH group to produce the inactive 3-0-phenolic glucuronide that is eliminated predominantly by the kidneys.[4] Ativan’s recorded half-life is 12-14 hours, and it has a clearance rate of 5.8 ml/min/kg.[4]
Ativan for Treatment
Anxiety disorders have been reported as the most common mental illness in the U.S., occurring in 18.1% of the population each year.[8] These disorders are treatable, and Ativan is one of the typical drugs used in the treatment of anxiety conditions. Ativan is used to treat anxiety by managing or giving short-term relief of the symptoms related to the disorder.[1] This can also be used in patients with depressive symptoms accompanying their anxiety.[1]
It also works well as an anticonvulsant and a complement to antipsychotics when treating mania and acute agitation.[9]
The indication for which Ativan is used will determine the dose prescribed.[9]
Side Effects of Ativan
Ativan can cause a range of adverse effects, mainly relating to depression of both the central nervous system (CNS) and respiratory system. These adverse effects are directly proportional to both dosage and age.[1]
Unfortunately, Ativan has the ability to worsen depressive disorders, or even bring previously undetected depression to the front.[1] This can explain why suicide ideation may occur in some patients and why benefits versus risks should be taken into account by the physician prescribing the drug, especially in patients who have a higher risk.[9]
Ativan also has the ability to make the patient dependant, both psychologically and physically.[1]
Mechanism of Action
Benzodiazepines produce their effect by binding to a certain site on the ligand-gated chloride (Cl–) sensitive GABAA receptors in the CNS.[9] This increases the rate at which Cl– channels open in postsynaptic neurons when the inhibitory neurotransmitter, gamma-aminobutyric acid (GABA), binds to its own receptor.[10] The massive influx of Cl– ions into the neuron results in hyperpolarization, therefore making it less excitable and resulting in the sedative and calming effects observed in patients. Due to the fact that Benzodiazepines bind to a different receptor than GABA, and they enhance its effects, Benzodiazepines are referred to as positive allosteric modulators.[9] Thus they do not function to replace or increase the affinity for GABA, but rather enhance the effects produced by the neurotransmitter.
GABA is found in the limbic system and the cerebral cortex in high concentrations and is one of the main neurotransmitters that regulate mood.[9] The GABA receptor that Benzodiazepines bind to is the GABAA receptor, which has three binding sites; of which two are for GABA and the other is where the drug binds.[9] This binding site is located in a pocket at the point where the α and γ subunits meet, thus the type of subunits present is vital in distinguishing the sensitivity and selectivity of the drug, as well as what side effects could potentially be expected.[11]
The α2 isoform observed in Benzodiazepine 2 (BZ2) receptors are responsible for the anxiolytic, and largely for the myorelaxant, effects of the drug.[9] These receptors are located in the dorsal horn of the spinal cord, motor neurons and the limbic system in high concentrations.[9] It is thought that the receptors found in the limbic system are responsible for the anxiolytic effects; whereas those located in the motor neurons and spinal cord produce the myorelaxant effects.
Anxiolytic Effects in Mice
The anxiolytic effects of Ativan and another drug were studied by Bagewadi et al.[12] They used a BALB/C strain of swiss albino mice and tested them with an Open Field Test and a Passive Avoidance Response. The Passive Avoidance box had a shock zone (SZ) consisting of a grid floor with an electronic shock of 20mV and a shock-free zone (SFZ) in the center.
The control group consisted of twelve mice who received a 10 ml/kg dosage of saline, and the Ativan group consisted of twelve mice who received a 0.5 mg/kg dosage of Ativan.[12] The mice were then tested on experimental day one, which was twenty-four hours following the first exposure, and again on experimental day eight.
In the Avoidance test, the three parameters used were:
- Step-down error: How many times the subject attempts to return to the SFZ.
- Step-down latency: The period of time the subject remains in the SFZ to avoid an electronic shock.
- Total time spent in the SZ
Results from the first day of the Avoidance test indicated a significant decrease in total time spent in the SZ and a decrease in the step-down latency and step-down error seen in the Ativan group compared to the control group.[12] Similar results were recorded for step-down latency on day eight; however, an increase in total time spent in the SZ and step-down error was observed.[12]
During the Open Field Test evaluation, the researchers reported that the Ativan group spent more time in the central square, crossed more squares, and had a decrease in freezing time compared to the control group.[12]
The results of both tests indicate significant anxiolytic effects of Ativan.
Tolerance to Ativan’s Anxiolytic Effects
Tolerance to Ativan is a topic much debated. Tolerance refers to the diminished response of a patient’s body to the effects of a drug, which is due to repeated use of the drug.[13]
Both File et al. [14] and Bourin et al. [15], using an Elevated Plus Maze and the Four Plates Test (Aron’s Test), respectively, observed no notable tolerance to Ativan’s anxiolytic effects in mice. In fact, it appeared that the anxiolytic effects were more pronounced with repeated administration when compared to a single dose.
It is important to keep in mind that tolerance observed in the mice may differ from that observed clinically since the half-lives of drugs in animals are shorter than treatment intervals.[15]
Sedative Effects in Mice
Sedation is a common side effect experienced by patients taking Ativan, a quality that has been used to treat patients for insomnia. One disadvantage of this treatment method is the development of tolerance and that abruptly stopping long-term treatment can result in rebound insomnia worse than the original issue.[16]
Lister et al. [17] set out to investigate tolerance to Ativan’s effects on sedation. Forty-nine male hooded rats were divided into the following seven groups:
- Control: Received daily injections containing the Ativan vehicle throughout the experiment.
- Acute: Received daily injections containing the Ativan vehicle for three days preceding the experiment, and then a dosage of 0.25 mg/kg Ativan twenty-five minutes before the Hole Board test.
- Chronic (0.125): Daily injections containing a dosage of 0.125 mg/kg Ativan.
- Chronic (0.25): Daily injections containing a dosage of 0.25 mg/kg Ativan.
- Chronic (0.50): Daily injections containing a dosage of 0.50 mg/kg Ativan.
- Sodium Pentobarbitone: Daily injections containing a dosage of 20 mg/kg sodium pentobarbitone.
- Ativan 0.5, 48hr: Three injections, each containing a dosage of 0.50 mg/kg Ativan separated by 48 hours.
Each group received their respective treatments for three days preceding the experiment. Twenty-four hours (forty-eight hours for the Ativan 0.5, 48hr group) following the last injection, each rat was injected with 0.25 mg/kg Ativan for a fourth time, except for the control group who received the Ativan vehicle.[17] After twenty-five minutes, each rat was given a chance to be placed in the Hole Board for 5 minutes.
Firstly, the researchers observed that the acute group experienced less time head-dipping and had overall less locomotor activity when compared with the control group. Thus, indicating the sedative effect of Ativan.[17]
When comparing the three chronic groups with the acute group, the researchers determined that the rats in the chronic groups experienced significantly greater locomotor activity and head-dipping. This result indicates tolerance for Ativan’s sedative effect.[17]
The comparison between the three chronic groups showed no significant difference in their effects on locomotor activity and head-dipping. This indicates that the degree of tolerance observed does not necessarily depend on the dosage received.[17],[18]
The same degree of tolerance observed in the above groups was also reported in the Ativan 0.5, 48hr group, further indicating that the resulting tolerance is not related to the dosage received since the animals experienced tolerance whether they received Ativan once every two days or once daily.[17]
No tolerance was observed in the sodium pentobarbitone group, which led the researchers to suggest that the tolerance exhibited was physiological and unrelated to learned behavior.[17]
The Myorelaxant Effect of Ativan
Bourin et al. [15] also investigated the effects Ativan has on myorelaxation.
They made use of a Rotarod Test to detect the motor function change that would be induced by Ativan’s myorelaxant effects. They set the Rotarod to 12 turns per minute and recorded the number of falls over three minutes. The researchers determined that myorelaxation was achieved by both single and repeated treatment of Ativan. Of the four benzodiazepines investigated, Ativan was one of the drugs that elicited a myorelaxant effect at lower dosages (a 0.25mg/kg dose). Still, it also appeared to be the least effective on myorelaxation.[15]
The researchers also noted that Ativan produced its sedative effect before its myorelaxant in the single-dose trial; however, this trend was reversed in the repeated administration trials with the myorelaxation occurring at 0.06 mg/kg and sedation at 0.125 mg/kg.
Since the mice exhibited myorelaxation after repeated administration, Bourin et al.[15] reported no observable tolerance to these effects. However, previous studies have observed minimal tolerance to Ativan’s myorelaxant effects.[19]
Conclusion
Ativan has shown promising results in its anxiolytic and myorelaxant abilities and even in the treatment of troubled sleep. These effects of Ativan in rodents were discussed, with tolerance being highlighted.
Animal models give a good indication of the effects of a drug on behavior. Studies using rodents have assisted researchers in determining the effects of Ativan and assisting them to establish the best treatment plan to get the desired effect without any/limited drawbacks (tolerance) in humans.
References
- “Ativan” RxList, https://www.rxlist.com/ativan-drug.htm
- “Ativan FDA Label” AccessData, https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/017794s044lbl.pdf
- “Lorazepam (Ativan)” AtivanDrugProject, https://ativandrugproject.weebly.com/structure-and-history.html
- “Lorazepam” Drugbank, https://go.drugbank.com/drugs/DB00186
- Batlle, E., Lizano, E., Vinas, M. and Pujol, M.D. 1,4-Benzodiazepines and New Derivatives: Description, Analysis, and Organic Synthesis. Medicinal Chemistry (2018) doi: 10.5772/intechopen.79879.
- “Lorazepam Injection USP” Pfizer, https://www.pfizer.ca/sites/default/files/201903/Lorazepam_PM_E_224238_26Feb2019.pdf
- Donaldson, M., Gizzarelli, G. and Chanpong, B. Oral Sedation: A Primer on Anxiolysis for the Adult Patient. Anesth Prog 54.3 (2007): 118-129
- “Facts and Statistics” ADAA, https://adaa.org/about-adaa/press-room/facts-statistics#:~:text=Anxiety%20disorders%20are%20the%20most,of%20those%20suffering%20receive%20treatment.
- Griffin, C.E., Kaye, A.M., Bueno, F.R. and Kaye, A.D. Benzodiazepine Pharmacology and Central Nervous System-Mediated Effects. The Ochsner Journal 13.2 (2013): 214-223
- “How Drugs Affect Neurotransmitters” TheBrain, https://thebrain.mcgill.ca/flash/i/i_03/i_03_m/i_03_m_par/i_03_m_par_benzodiazepines.html
- Kessler, R.C., McGonagle, K.A., Zhao, S., Nelson, C.B., Hughes, M., Eshleman, S., et al. Lifetime and 12-month prevalence of DSM-III-R psychiatric disorders in the United States: Results from the National Comorbidity Survey. Archives of General Psychiatry 51.1 (1994): 8-19
- Bagewadi, H.G., Ak, A.K. and Shivaramegowda, R.M. An Experimental Study to Evaluate the Effect of Memantine in Animal Models of Anxiety in Swiss Albino Mice. J Clinical and Diagnostic Research 9.8 (2015): FF01-FF05
- “Tolerance and Resistance to Drugs” MSD Manual, https://www.msdmanuals.com/home/drugs/factors-affecting-response-to-drugs/tolerance-and-resistance-to-drugs#:~:text=Tolerance%20is%20a%20person’s%20diminished,drug%20usually%20effective%20against%20them.
- File, S.E., Wilks, L.J. and Mabbutt, P.S. Withdrawal, Tolerance and Sensitization After a Single Dose of Lorazepam. Pharm, Biochem and Behav 31 (1989): 937-940
- Bourin, M., Hascoet, M., Mansouri, B., Colombel M.C. and Bradwejn, J. Comparison of Behavioural Effects After Single and Repeated Administrations of Four Benzodiazepines in Three Mice Behavioural Models. J Psychiatr Neurosci 17.2 (1992): 72-77
- “Overcoming Insomnia” Harvard Health Publishing, https://www.health.harvard.edu/newsletter_article/overcoming-insomnia#:~:text=In%20practice%2C%20many%20of%20the,feel%20refreshed%20the%20next%20morning.
- Lister, R.G., File, S.E. and Greenblatt, D.J. Functional Tolerance to Lorazepam in the rat. Psychopharmacology 81 (1983): 292-294
- Lister, R.G., File, S.E. and Greenblatt, D.J. The Behavioural Effects of Lorazepam are Poorly Related to Its Concentration in the Brain. Life Sciences 32.17 (1983) 2033-2040
- Miller, L.G., Woolwerton, S., Greenblatt, D.J., Lopez, F., Roy, R.B. and Shader, R.I. Chronic benzodiazepine administration. IV. Rapid development of tolerance and receptor down regulation associated with alprazolam administration. Biochem Pharmacol 38 (1989): 3773-3777