
Implantable Osmotic Release Pump
Ensures continuous and uninterrupted administration, maintenance, and constant administration of a drug to an animal. Storage capsules have good drug compatibility and protection for short half-life drugs. Capsules have good biocompatibility and d...
| capacity_options | 100 µl, 200 µl, 2 ml |
| flow_regulator_tube_material | 304 stainless steel |
| syringe_type | Flat head for injection of liquid |
| reservoir_wall_material | polyester |
| delivery_duration_range | one day to a month and a half |
| usage | single use only |
Specifications
|
Model |
Capacity |
Duration |
Pumping Rate(μL/hr) |
Flow Regulator* Cap |
|---|---|---|---|---|
|
RWD-1003D |
100μl |
3D |
1 |
Without |
|
RWD-1001W |
100μl |
1W |
0.5 |
Without |
|
RWD-1002W |
100μl |
2W |
0.25 |
Without |
|
RWD-1004W |
100μl |
4W |
0.125 |
Without |
|
RWD-1006W |
100μl |
6W |
0.083 |
Without |
|
RWD-2003D |
200μl |
3D |
2 |
PE material |
|
RWD-2001W |
200μl |
1W |
1 |
PE material |
|
RWD-2002W |
200μl |
2W |
0.5 |
PE material |
|
RWD-2004W |
200μl |
4W |
0.25 |
PE material |
|
RWD-2ml1W |
2ml |
1W |
10 |
PE material |
|
RWD-2mL2W |
2ml |
2W |
5 |
PE material |
|
RWD-2ml4W |
2ml |
4W |
2 |
PE material |
*Flow regulator_Tube: 304 stainless steel material. Infusion_Syringe:Flat head for the injection of liquid.
Introduction
The implantable osmotic release pump is an inexpensive cutting-edge drug delivery system. You can use this small implantable infusion pump for preclinical pharmaceutical research in mice, rats, and other laboratory animals. The mini-pump delivers drugs, hormones, and other test compounds at continuous and controlled rates, for terms extending from one day to a month and a half, without the requirement for external interference. The implantable pumps utilize osmosis for continuous infusion of unrestrained laboratory animals.
In the previous three decades, drug delivery research has made critical headways because of the recent developments and innovations in the fields of pharmaceutical sciences including pharmacokinetics, pharmacodynamics, and biopharmaceutics. Low development costs and regulated drug delivery pushed the research. Hence a significant portion of the novel drug delivery frameworks is upgraded such that the drug dose and dosing interim are reduced thereby maintaining optimum therapeutic dose and efficacy. These innovative drug delivery systems have been designed to regulate drug release over an extended time frame. Furthermore, recent advances have been made to make the rate and extent of the drug release independent of physicochemical properties of drugs and excipients, and physiological factors like pH of the gastrointestinal tract, the presence of food, and nutritional health.
Osmotic pumps are the most promising procedure based systems for controlled drug delivery. The controlled drug delivery system is mediated by osmosis which can be characterized as the net movement of water molecules over a selectively permeable film driven by a difference in osmotic gradient over the layer. The difference in solute concentration over the membrane permits entry of water, however, rejects most solute particles. Osmogens create osmotic pressure to stimulate drug release from the pump.
Apparatus And Equipment
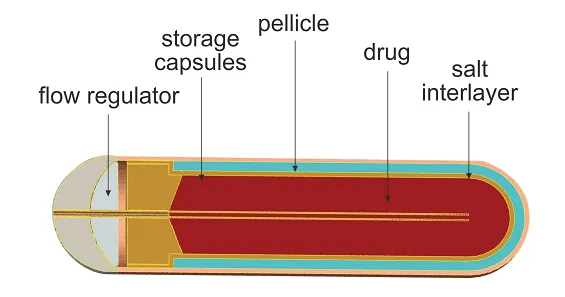
In cross-section, the implantable osmotic release pumps are composed of a drug core (reservoir), the diffusion agent, and the tissue layer (rate controller). Additionally, a flow moderator is inserted into the body of the diffusion pump. It is a kind of an implantable system within which you can load a solution or suspension contained in a cylindrical reservoir shaped from an artificial collapsible, impervious stuff wall (e.g., polyester) that is open to the external surroundings via a single orifice.
The main components of an implantable osmotic release pump include the drug compound (water-soluble or insoluble), osmotic agents (ionic compounds of inorganic salts), a stable semi-permeable membrane, plasticizers, flux regulators, wicking agents, pore forming agent, and coating solvent.
Our implantable osmotic release pump is available in three different sizes to offer the researchers a variety of dosing capacities, release rates, and duration. You can load it with 100 µl, 200 µl, and 2 ml of the test compound.
The implantable osmotic pump employs an osmotic gradient inside the lumen called the salt sleeve and the tissue condition in which the pump is implanted. The high osmolality of the salt sleeve makes water flow into the pump through a semipermeable film which encases the external surface of the pump. As the water enters the salt sleeve, it compresses the flexible reservoir, dislodging the test substance from the tube at a controlled rate. The rate of drug delivery is directly proportional to the osmotic pressure of the core. Since the delivery system cannot be refilled, these pumps are intended for single use only.
The rate of the drug delivery by our osmotic pump is controlled by the water penetrability from the pump’s external film. In this manner, the drug delivery is independent of the drug formulation and excipients. Drugs of different atomic arrangements, including ionized medications and macromolecules, can be administered persistently at controlled rates. The sub-atomic weight of a compound, or its physical and chemical properties, does not affect the rate of the drug delivery by the implantable osmotic release pumps.
Procedure
The osmotic pumps can be implanted subcutaneously or intraperitoneally depending on the size of the animal. For targeted drug delivery, a catheter can be attached to the osmotic pump to gain access to the tissues of interest. Subcutaneous implantation is technically the easiest and least intrusive procedure. Follow these steps for subcutaneous implantation:
- Anesthetize the animal with ketamine or any other rodent anesthetizer.
- Shave and wash the skin over the implantation site.
- Make an incision adjacent to the implantation site. If the implantation is planned on the backside of the animal, then the mid-scapular incision is made.
- To avoid excessive blood flow, place a hemostat onto the incision. Make an appropriate pocket for the pump by manipulating the subcutaneous tissues by hemostat. Keep in mind that the pocket should be large enough to allow the pump movement.
- Insert the pump containing the drug into the pocket in the incision.
- With the help of incision clips, close the wound.
The osmotic system can also be implanted in the peritoneal cavity of the larger animals. Follow the below-mentioned protocol for intraperitoneal injection:
- After anesthetizing the animal, shave and wash the skin over the implantation site.
- In the lower abdomen, make a 1 cm long midline skin incision.
- Tent the peritoneal muscles and carefully incise the peritoneal wall keeping the bowel safe.
- Insert the osmotic pump into the cavity.
- Close the abdomen with the help of sutures and incision clips.
Keep in mind that the pumps should be explanted if the animals survive after active infusion. The pumps must be removed no later than the first half-life of the test compound. The pump should be removed to measure the residual volume to confirm delivery, verify a drug’s stability, and assure the bioactivity of the test compound.
Applications
Implantable consistent infusion osmotic pumps have many applications in preclinical drug delivery improvement. The system empowers continuous and controlled dosing permitting the accomplishment of enduring state conditions and precise drug delivery. The system offers the researchers temporal and spatial control over the drug release. It circumvents poor-availability hurdles in the challenging therapeutic studies of hormones and growth factors. Also, the drugs with faster clearance rates and shorter half-lives require a consistent dosage that can be easily achieved by the implantable osmotic release pumps. Furthermore, it can assist the drug delivery of the compounds having a lower therapeutic index by continuous infusion while avoiding toxic concentrations. Moreover, the osmotic systems not only deliver the drugs with moderate solubility but also with extreme solubility.
Continuous infusion of the therapeutic compounds with the help of the implantable osmotic release pump assists the experimenters to establish parameters of the drugs with unknown pharmacokinetics. These pumps can also be used for a comparative study of the efficacy of different drugs administered through different routes. In addition to the drug efficacy analysis, these drug delivery systems can also be used for the targeted delivery of chemotherapeutic agents to the tumors with the help of an attached catheter. The osmotic release pumps can not only be used for chemotherapeutic preclinical studies but also to monitor cell proliferation to assess the carcinogenic potential of the cells. Also, these pumps improve bio-luminescence imaging studies by continuously delivering bio-luminescent substrates.
Applications of the osmotic release pump include the disciplines of oncology, stem cell research, gene transfection, gene silencing, neuroscience research, and preclinical pharmacological studies.
Strengths And Weaknesses
- The osmotic release pump ensures round-the-clock delivery of the test molecules at a controlled rate.
- Zero-order drug release after an initial tag is one of the significant advantages of the implantable osmotic release pump.
- The tool offers drug delivery independent of physiological factors like gastric health, pH, and hydrodynamic conditions.
- The drug release can be easily controlled by regulating the amount of water filled in the pump.
- The equipment permits continuous and regulated administration of short peptides and proteins.
- It offers a convenient method for the persistent dosing of laboratory animals.
- It minimizes unwanted experimental variables and makes sure reproducible, constant results.
- It eliminates the need for nighttime or weekend dosing.
- Its small size increases its popularity for use in mice and small rats.
- The osmotic drug delivery system allows for focused and targeted drug administration.
- It can be used for both in-vivo and in-vitro studies.
- It provides the biomedical researchers with cost-effective research tool.
- There may be a chance of dose dumping if the film coating is not correct.
- The size of the orifice is critical for controlled drug release.
- It may cause ulceration or irritation at the site of implantation.
Osmotic pumps are one of the novel pharmaceutical tools for controlled and consistent drug delivery. Osmotic drug delivery pumps commonly comprise a drug center containing osmogen that is covered with a semipermeable layer. This covering has at least one transportation ports through which the test compound or suspension of the medication is discharged after some time. Different endeavors are underway to make an effective osmotic drug delivery system like pulsatile delivery with an expandable hole, and a lipid osmotic pump containing a smaller capsules than the usual osmotic pump for continuous and extended-release.
References
- Continuous Drug Infusion Model with an Implantable Osmotic Pump. Retrieved from: https://noblelifesci.com/uploads/file/Noble%20Life%20SciencesContinuous%20Infusion%20Model.pdf
- Keraliya, R. A., Patel, C., Patel, P., Keraliya, V., Soni, T. G., Patel, R. C., & Patel., M. M. (2012). Osmotic Drug Delivery System as a Part of Modified Release Dosage Form. ISRN Pharmaceutics.
- Mathur, M., & Mishra, R. A review on osmotic pump drug delivery system. International journal of pharmaceutical sciences and research.
- Popesko, P., et al. (1992) A color atlas of small laboratory animals, Volume Two, Rat, Mouse & Hamster, London: Wolf Publishing Ltd.
- Prescott LF. Novel Drug Delivery and Its Therapeutic application. West Susset, UK: John Wiley and Sons; 1989. The need for improved drug delivery in clinical practice; pp. 1–11.
- Stepkowski, S. M., Tu, Y., Condon, T. P., Bennett, C. F. (1994) ‘Blocking of heart allograft rejection by intercellular adhesion molecule-1 antisense oligonucleotides alone or in combination with other immunosuppressive modalities‘, Journal of Immunology, 153, 5336-5346.
- Tu, Y., Stepkowski, S. M., Chou, T.-C., Kahan, B. D. (1995) ‘The synergistic effects of cyclosporine, sirolimus, and brequinar on heart allograft survival in mice,’ Transplantation, 59(2), 177-183.
How It Works
The osmotic pump operates through osmotic pressure differential across a semi-permeable membrane. When implanted, body fluids penetrate the membrane and dissolve the osmotic salt tablet within the pump core. This creates an osmotic gradient that forces the drug solution from the reservoir through the delivery orifice at a predetermined rate. The polyester reservoir wall maintains structural integrity while allowing precise volumetric delivery.
Flow rate is controlled by the membrane permeability, orifice diameter, and osmotic driving force, remaining constant regardless of implantation site or physiological conditions. The 304 stainless steel delivery system ensures biocompatibility and prevents flow obstruction during chronic implantation periods. Unlike pulsatile injection methods, this mechanism provides zero-order kinetics with minimal plasma concentration fluctuations.
Features & Benefits
Duration
- 3D
- 1W
- 2W
- 4W
- 6W
capacity_options
- 100 µl, 200 µl, 2 ml
flow_regulator_tube_material
- 304 stainless steel
syringe_type
- Flat head for injection of liquid
reservoir_wall_material
- polyester
delivery_duration_range
- one day to a month and a half
usage
- single use only
delivery_method
- osmotic gradient driven
membrane_type
- semi-permeable
Capacity
- 2mL
- 100µL
- 200µL
Material
- 304 stainless steel
- polyester
Species
- Mouse
- Rat
Brand
- RWD
Research Domain
- Addiction Research
- Behavioral Pharmacology
- Cancer Research
- Cardiovascular Research
- Metabolic Research
- Neuroscience
- Pain Research
- Toxicology
Weight
- 8.27 lbs
Dimensions
- L: 34.0 in
- W: 39.0 in
- H: 33.0 in
Comparison Guide
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Reservoir Capacity Range | 100 μL to 2 mL across model series | Many systems offer narrower capacity ranges or require different platforms | Single platform accommodates diverse experimental scales from acute studies to chronic protocols. |
| Flow Rate Precision | 0.083-10 μL/hr with osmotic control mechanism | Battery-powered pumps may show flow drift over time | Osmotic mechanism maintains consistent delivery without power source degradation. |
| Delivery Duration Options | 3 days to 6 weeks in single platform | Fixed duration pumps often require separate models for different time points | Flexible study design within standardized surgical and analytical protocols. |
| Construction Materials | 304 stainless steel flow components with polyester reservoir | Lower-grade materials may affect biocompatibility or flow stability | Medical-grade materials ensure consistent performance in physiological environments. |
| Priming Requirements | 4-6 hour priming period at 37°C | Some systems require longer equilibration or complex priming procedures | Rapid study initiation with predictable flow onset timing. |
This osmotic pump series provides a comprehensive platform for controlled drug delivery research, offering multiple capacity and duration options within a single biocompatible design. The osmotic mechanism ensures consistent flow rates without external power requirements, supporting reproducible pharmacokinetic studies across diverse experimental protocols.
Practical Tips
Load pumps completely to eliminate air bubbles, which can disrupt osmotic flow and cause variable delivery rates.
Why: Air bubbles create compressible spaces that interfere with the precise pressure-driven delivery mechanism.
Verify flow rates in vitro using the same temperature and solution conditions planned for in vivo studies.
Why: Solution viscosity and temperature significantly affect flow through the delivery orifice.
Inspect delivery orifice for blockages before implantation using low-power microscopy or flow testing.
Why: Microscopic debris or manufacturing residue can completely obstruct drug delivery.
Document exact implantation depth and anatomical location for each animal to enable flow rate correlation analysis.
Why: Tissue pressure and vascular proximity can influence effective drug distribution patterns.
If flow appears reduced, check for fibrous capsule formation around the delivery orifice at necropsy.
Why: Host tissue response can create barriers that impede drug diffusion from the pump exit.
Use appropriate containment and disposal protocols for pumps containing controlled substances or hazardous compounds.
Why: Residual drug content may pose exposure risks during pump retrieval and disposal procedures.
Plan drug loading procedures to minimize exposure time between filling and implantation, especially for light-sensitive compounds.
Why: Extended exposure to laboratory conditions can degrade drug potency before delivery begins.
Weigh pumps before and after use to validate actual drug delivery volume against theoretical calculations.
Why: Direct measurement provides the most accurate assessment of pump performance for pharmacokinetic modeling.
Setup Guide
What’s in the Box
- Osmotic pump (capacity and duration as specified in model selection)
- Flat-head infusion syringe
- 304 stainless steel flow regulator tube
- PE material flow regulator cap (200 μL and 2 mL models)
- Sterile packaging
- Product specification sheet (typical)
- Implantation guidelines (typical)
Warranty
ConductScience provides a standard manufacturer warranty covering defects in materials and workmanship. Technical support includes implantation protocol guidance and troubleshooting assistance for optimal experimental outcomes.
Compliance
References
Background reading relevant to this product:
How do I calculate the required drug concentration for my target dose?
Multiply the desired dose rate (mg/kg/hr) by body weight, then divide by the pump flow rate (μL/hr) to determine required concentration (mg/mL). Account for drug stability over the delivery duration.
What factors affect pump flow rate accuracy in vivo?
Temperature variations, implantation depth, and tissue encapsulation can influence flow rates. Maintain consistent implantation technique and consider in vitro flow rate verification before implantation.
Can I use these pumps for intracerebroventricular delivery?
Yes, connect the delivery orifice to an appropriate cannula system. Ensure cannula patency and proper cerebrospinal fluid flow dynamics for accurate drug targeting.
How should I handle drug stability concerns during extended delivery?
Validate drug stability at 37°C over the intended delivery duration. Consider antioxidants or stabilizing excipients, and verify biological activity at study endpoint.
What is the priming time required before implantation?
Prime loaded pumps in sterile saline at 37°C for 4-6 hours to eliminate initial lag time and establish steady-state flow before surgical implantation.
How do I verify complete drug delivery at study termination?
Retrieve pumps post-mortem and measure residual volume. Compare with expected delivery based on flow rate and duration to confirm pump performance.
Are there size limitations for different species?
Consider animal size relative to pump dimensions and reservoir capacity. Smaller pumps (100 μL) are suitable for mice, while larger capacities accommodate rat studies requiring extended delivery periods.
Can I use organic solvents with these pumps?
Consult material compatibility data for specific solvents. The polyester reservoir and membrane may have limited compatibility with certain organic solvents that could affect pump integrity.
Have a question about this product?
Accessories
Enhance your setup with compatible accessories




