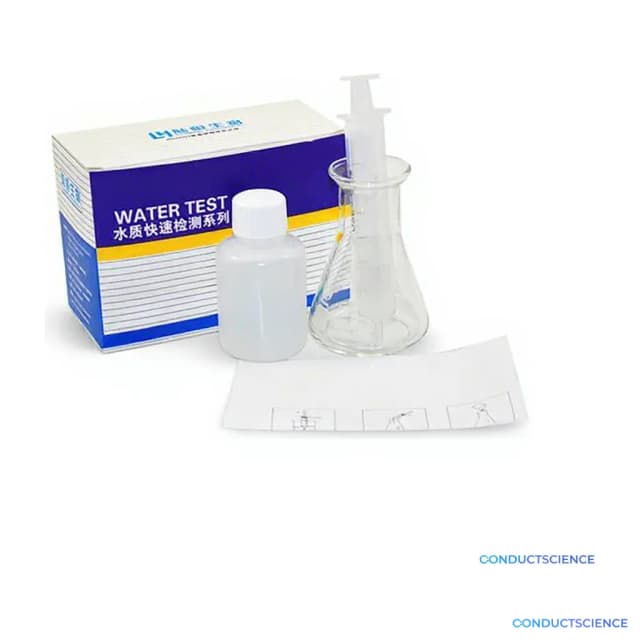
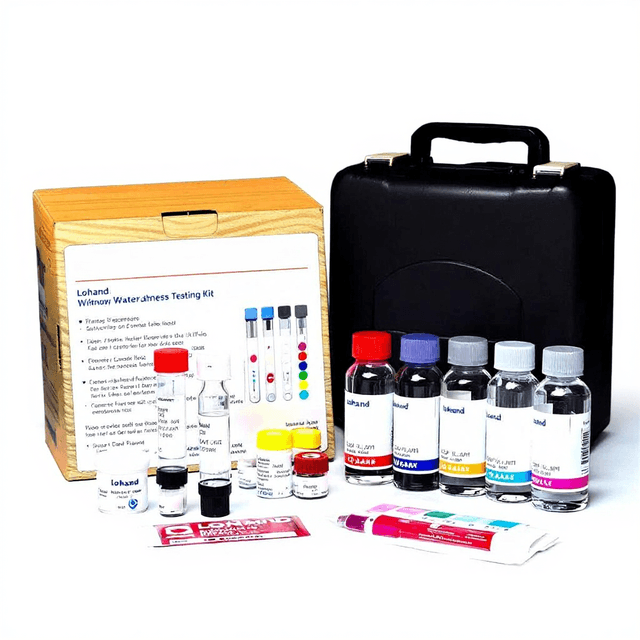
Hard Water Test Kit
Analytical test kit for quantitative measurement of water hardness through calcium and magnesium ion analysis using standardized titration methodology.

Louise Corscadden, PhD
Neuroscience · ConductScience
Ask Louise about Hard Water Test Kit fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
The Hard Water Test Kit (Model LH2011) provides quantitative measurement of water hardness through calcium and magnesium ion analysis. This analytical kit enables researchers to assess total hardness levels in aquatic samples using standardized titration methodology. The system supports water quality assessments across environmental monitoring, aquaculture research, and industrial water treatment applications.
Water hardness measurement is essential for characterizing aquatic environments, optimizing experimental conditions, and monitoring water chemistry parameters. The kit provides reliable quantification of dissolved calcium carbonate equivalents, supporting both field sampling protocols and laboratory-based water analysis workflows.
How It Works
Water hardness measurement relies on complexometric titration using EDTA (ethylenediaminetetraacetic acid) as the titrant. Calcium and magnesium ions form stable chelate complexes with EDTA in a 1:1 stoichiometric ratio. The endpoint is detected using a metal indicator that changes color when all divalent cations have been complexed with EDTA.
The titration is performed at pH 10 using an ammonia buffer to ensure complete complexation. Eriochrome Black T or similar indicators provide visual endpoint detection, transitioning from wine-red (metal-indicator complex) to blue (free indicator) at the equivalence point. Total hardness is calculated from the EDTA volume consumed and expressed as mg/L calcium carbonate equivalents.
This methodology provides quantitative analysis of total hardness without interference from monovalent cations, enabling accurate assessment of water mineral content relevant to aquatic research applications.
Features & Benefits
Product name
- Model No.
Hard water test kit
- LH2011
Automation Level
- manual
Brand
- ConductScience
Research Domain
- Analytical Chemistry
- Environmental Monitoring
Weight
- 0.26 kg
Dimensions
- L: 30.0 mm
- W: 20.0 mm
- H: 10.0 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Analytical Methodology | Complexometric titration with visual endpoint detection | Test strips often provide only semi-quantitative colorimetric results | Quantitative analysis enables precise documentation of water chemistry parameters for research protocols |
| Measurement Principle | EDTA complexation with standardized reagent system | Digital meters may require frequent calibration and maintenance | Chemical titration provides inherent analytical standards without electronic calibration requirements |
| Sample Analysis Format | Batch processing capability for multiple samples | Single-sample analyzers limit throughput capacity | Supports comprehensive water quality surveys and replicate analysis protocols |
| Analytical Precision | Standardized complexometric chemistry with established protocols | Varies by model and calibration maintenance | Consistent analytical performance based on fundamental chemical principles rather than instrument stability |
The kit provides quantitative hardness analysis through established complexometric titration methodology. Visual endpoint detection eliminates instrumentation requirements while maintaining research-grade analytical precision for water chemistry characterization.
Practical Tips
Verify EDTA titrant concentration using certified calcium standard solutions before critical analyses.
Why: Titrant standardization ensures accurate quantitative results and traceability to analytical standards.
Store indicators in dark containers and monitor for color changes that indicate reagent degradation.
Why: Light-sensitive indicators lose endpoint detection capability, compromising analytical accuracy.
Analyze samples promptly after collection to prevent calcium carbonate precipitation in alkaline waters.
Why: Mineral precipitation removes dissolved ions from solution, leading to underestimation of true hardness levels.
If endpoint detection is unclear, adjust lighting conditions or dilute highly colored samples before analysis.
Why: Visual endpoint detection requires clear color transition observation for accurate titration volume determination.
Run duplicate titrations and calculate relative percent difference to assess analytical precision.
Why: Replicate analysis identifies procedural errors and establishes confidence in quantitative results.
Handle ammonia buffer solutions in well-ventilated areas and avoid skin contact with reagents.
Why: Ammonia vapors can cause respiratory irritation and chemical reagents may cause skin sensitization.
Setup Guide
What’s in the Box
- EDTA titrant solution (typical)
- Buffer solution reagents (typical)
- Metal indicator solution (typical)
- Titration apparatus (typical)
- Sample measurement containers (typical)
- Analytical protocol instructions (typical)
- Calculation worksheets (typical)
Warranty
ConductScience provides standard warranty coverage for reagent quality and analytical performance specifications. Technical support available for methodology questions and troubleshooting.
Compliance
What is the detection limit and measurement range for total hardness analysis?
Consult product datasheet for specific detection limits and analytical range. Typical complexometric methods detect hardness levels from low mg/L to several hundred mg/L as CaCO3 equivalents.
How does sample pH affect the accuracy of hardness measurements?
The analysis requires pH 10 maintained by ammonia buffer to ensure complete EDTA complexation. Acidic samples may require additional buffer, while highly alkaline samples could affect indicator performance.
What interferences should be considered in natural water samples?
Heavy metals, high iron concentrations, and organic matter can interfere with endpoint detection. Sample pretreatment may be required for highly colored or metal-contaminated waters.
How should reagents be stored to maintain analytical performance?
Store reagents according to manufacturer specifications, typically in cool, dark conditions. EDTA solutions should be protected from metal contamination, and indicators require protection from light degradation.
What quality control measures are recommended for routine analysis?
Run duplicate analyses, analyze blank solutions, and use certified reference materials when available. Monitor reagent stability and endpoint detection consistency across analytical batches.
Can this kit be used for seawater or high-salinity samples?
High ionic strength samples may require method modifications or dilution. Consult methodology documentation for guidance on saline water analysis and appropriate sample preparation.
Have a question about this product?
Accessories
Enhance your setup with compatible accessories