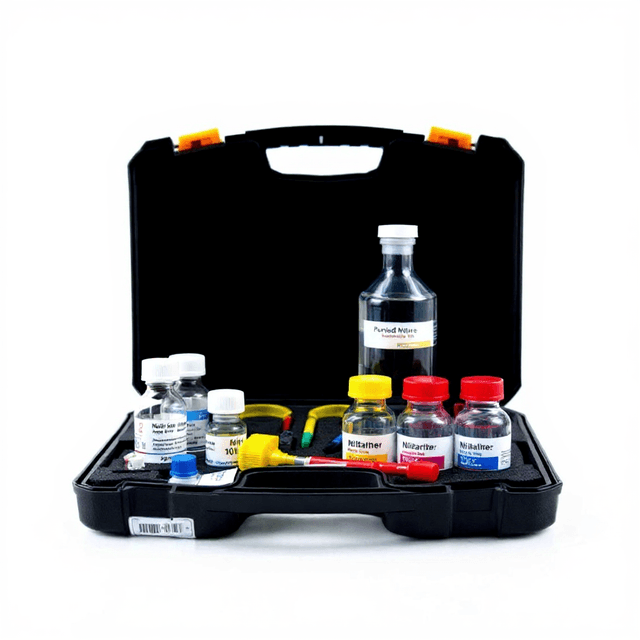
Chloride Test Kit
Silver nitrate titration kit for quantitative chloride determination in water samples, supporting environmental monitoring and water quality assessment applications.

Louise Corscadden, PhD
Neuroscience · ConductScience
Ask Louise about Chloride Test Kit fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
The Chloride Test Kit (Model LH2015) provides quantitative determination of chloride ion concentration in aqueous samples using silver nitrate titration methodology. This analytical kit enables field and laboratory assessment of chloride levels in water samples, supporting environmental monitoring, water quality assessment, and process control applications.
The kit employs the standard Mohr titration method, utilizing silver nitrate as the titrant and potassium chromate as the indicator. This classical analytical approach provides reliable quantitative results for chloride determination across a range of sample types, from natural waters to industrial process streams.
How It Works
The kit utilizes the Mohr titration method, a classical argentometric procedure for chloride quantification. Silver nitrate solution reacts stoichiometrically with chloride ions to form silver chloride precipitate according to the reaction: Ag⁺ + Cl⁻ → AgCl(s). The endpoint is detected using potassium chromate indicator, which forms a reddish-brown silver chromate precipitate when excess silver ions are present.
The titration is performed in neutral to slightly alkaline conditions (pH 6.5-10.5) to ensure optimal indicator performance. Sample preparation typically involves filtration to remove suspended solids and pH adjustment if necessary. The chloride concentration is calculated from the volume of silver nitrate consumed and the known normality of the titrant solution.
This method provides accuracy within ±2% for chloride concentrations above 10 mg/L, with detection limits suitable for most water quality applications. The visual endpoint determination makes this approach particularly valuable for field applications where instrumental analysis is not available.
Features & Benefits
Product name
- Model No.
chloride test kit
- LH2015
Automation Level
- manual
Brand
- ConductScience
Research Domain
- Analytical Chemistry
- Clinical Diagnostics
- Environmental Monitoring
- Food Science
- Industrial Hygiene
- Pharmaceutical QC
Weight
- 0.26 kg
Dimensions
- L: 30.0 mm
- W: 20.0 mm
- H: 10.0 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Analysis Method | Silver nitrate titration with visual endpoint | Ion chromatography or electrode methods often used for automated analysis | Provides reliable results without expensive instrumentation or electrical power requirements |
| Sample Throughput | Manual analysis suitable for moderate sample loads | Automated systems offer higher throughput capabilities | Enables careful analytical control and educational demonstration of classical methods |
| Field Portability | Complete portable kit with all reagents included | Instrumental methods typically require laboratory infrastructure | Supports on-site analysis for environmental monitoring and process control applications |
| Detection Principle | Visual color change endpoint detection | Electronic detection systems common in modern analyzers | Provides direct visual confirmation of analytical endpoint without calibration dependencies |
This chloride test kit offers classical analytical reliability with complete reagent systems for immediate field or laboratory use. The silver nitrate titration method provides established accuracy without requiring expensive instrumentation or electrical power.
Practical Tips
Standardize silver nitrate titrant daily using certified sodium chloride reference standard to ensure analytical accuracy.
Why: Titrant concentration can change due to light exposure and carbonate contamination affecting quantitative results.
Maintain consistent lighting conditions and use white background when observing endpoint color changes.
Why: Endpoint detection accuracy depends on visual observation of subtle color transitions from yellow to reddish-brown.
Store silver nitrate solution in amber bottles away from light to prevent photodecomposition.
Why: Light exposure causes silver reduction and formation of metallic silver, altering titrant concentration.
Run samples in duplicate and calculate relative percent difference to assess analytical precision.
Why: Duplicate analysis identifies pipetting errors and ensures consistent technique application.
If endpoint is difficult to detect, adjust sample pH to 7-8 range and ensure adequate indicator concentration.
Why: Indicator performance is pH-dependent and insufficient chromate leads to unclear endpoint transitions.
Handle silver nitrate with appropriate PPE as it causes skin staining and is a strong oxidizing agent.
Why: Silver nitrate contact with organic materials can cause burns and permanent discoloration of skin and clothing.
Filter turbid samples before analysis to prevent interference with endpoint visibility.
Why: Suspended particles can mask the color transition and lead to premature or delayed endpoint detection.
Setup Guide
What’s in the Box
- Silver nitrate titrant solution (typical)
- Potassium chromate indicator solution (typical)
- Sodium chloride reference standard (typical)
- pH adjustment buffers (typical)
- Graduated burette and stand (typical)
- Volumetric pipettes (typical)
- Analytical protocol and calculation guide (typical)
- Safety data sheets for all reagents (typical)
Warranty
ConductScience provides standard warranty coverage for reagent quality and kit completeness, with technical support available for analytical methodology and troubleshooting assistance.
Compliance
What is the analytical range and detection limit for chloride determination?
The kit typically provides quantitative results for chloride concentrations from 10-1000 mg/L with ±2% accuracy. Detection limits depend on sample volume and titrant normality - consult product datasheet for specific analytical parameters.
How do interfering ions affect the analysis accuracy?
Bromide and iodide ions interfere positively as they also precipitate with silver nitrate. Sulfide, sulfite, and thiosulfate interfere negatively. Sample pretreatment or alternative methods may be required for samples containing significant interferents.
What sample preservation and storage requirements apply?
Chloride samples are chemically stable and require no preservation for most applications. Samples should be stored in clean containers at room temperature and analyzed within reasonable time to prevent contamination.
Can this method be used for seawater and high-salinity samples?
Yes, but high-chloride samples may require dilution to bring concentrations within the analytical range. Use appropriate dilution factors in final calculations and ensure adequate endpoint visibility.
What quality control procedures should be implemented?
Run duplicate analyses, blank corrections, and periodic standardization checks. Include certified reference materials when available and maintain control charts for analytical precision monitoring.
How does this compare to ion chromatography or electrode methods?
Titration provides excellent accuracy for routine chloride determination without expensive instrumentation. Ion chromatography offers better selectivity and automation, while ion-selective electrodes provide faster results but may have interference issues.
Have a question about this product?
Accessories
Enhance your setup with compatible accessories