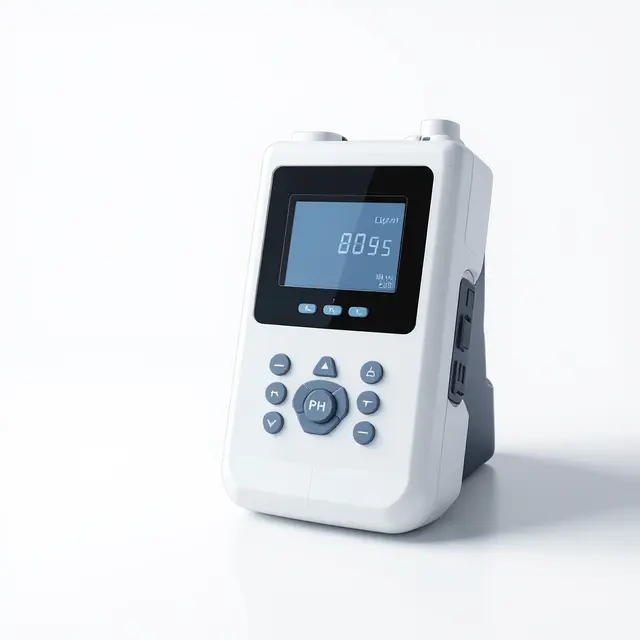
Benchtop Dissolved Oxygen Meter DO400F
Benchtop dissolved oxygen meter measuring 0.00-99.99 ppm with temperature compensation, barometric correction, and GLP-compliant data management features.

Louise Corscadden, PhD
Neuroscience · ConductScience
Ask Louise about Benchtop Dissolved Oxygen Meter DO400F fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
The Benchtop Dissolved Oxygen Meter DO400F provides precise measurement of dissolved oxygen concentration in aqueous solutions with a range of 0.00-99.99 ppm. This laboratory instrument features automatic and manual temperature compensation, barometric pressure compensation, and manual salinity factor correction to ensure accurate readings across varying environmental conditions. The meter incorporates GLP-compliant data management features and supports calibration using air-saturated water or zero point standards.
Designed for laboratory and field applications requiring reliable dissolved oxygen monitoring, the DO400F offers both automatic temperature compensation (ATC) and manual temperature compensation (MTC) modes. The instrument provides clear reading prompts indicating measurement status (reading, stable, locked) and includes data storage capabilities for documentation and quality assurance protocols.
How It Works
The DO400F utilizes an electrochemical dissolved oxygen sensor to measure oxygen concentration in aqueous solutions. The sensor typically employs a polarographic or galvanic cell design where oxygen diffuses through a permeable membrane and undergoes reduction at the cathode, generating a current proportional to the dissolved oxygen concentration.
Temperature compensation is critical for accurate dissolved oxygen measurements because oxygen solubility decreases with increasing temperature. The instrument provides both automatic temperature compensation (ATC) using an integrated temperature probe and manual temperature compensation (MTC) for applications where external temperature measurement is preferred. Barometric pressure compensation accounts for atmospheric pressure variations that affect oxygen saturation levels, while salinity correction adjusts for the reduced oxygen solubility in salt solutions.
The meter's calibration system uses air-saturated water as the reference standard, representing 100% oxygen saturation at the measured temperature and pressure conditions. Zero point calibration can be performed using oxygen-free solutions prepared with sodium sulfite or nitrogen sparging to establish the baseline measurement.
Features & Benefits
Automation Level
- manual
DO Concentration
- Sensor Type
Range
- 0.00~99.99ppm
Temperature Range
- Range
Calibration Points
- Air-saturated water or zero point
Manual Salinity Factor Correction
- Yes
Barometric Compensation
- Yes
DO Saturation
- Range
Measurement
- Reading Mode
Reading Prompts
- Reading, stable, locked
Temp. Compensation
- ATC, MTC
Data Management
- Data Storage
GLP Features
- Yes
Brand
- ConductScience
Research Domain
- Analytical Chemistry
- Clinical Diagnostics
- Environmental Monitoring
- Food Science
- Microbiology
- Pharmaceutical QC
Accuracy
- ±0.1℃
Weight
- 0.9 kg
Dimensions
- L: 6.8 mm
- W: 24.2 mm
- H: 19.5 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Measurement Range | 0.00-99.99 ppm dissolved oxygen concentration | Entry-level models often provide narrower ranges or lower resolution | Wide range covers from anaerobic to supersaturated conditions in diverse research applications. |
| Temperature Compensation | Both automatic (ATC) and manual (MTC) temperature compensation modes | Basic meters typically offer only automatic compensation | Flexibility to use external temperature references when probe-based measurement is not suitable for the sample matrix. |
| Environmental Corrections | Barometric pressure compensation and manual salinity factor correction | Lower-cost models often lack barometric and salinity corrections | Enhanced accuracy for field measurements and samples with varying ionic strength or at different altitudes. |
| Data Management | GLP-compliant data storage with reading status indicators | Basic models may lack data storage or compliance features | Supports regulatory documentation requirements and quality assurance protocols in controlled laboratory environments. |
| Calibration Options | Air-saturated water or zero point calibration standards | Varies by model - some require proprietary calibration solutions | Uses easily prepared calibration standards that can be made in-house, reducing ongoing operational costs. |
The DO400F offers comprehensive dissolved oxygen measurement capabilities with dual temperature compensation modes, environmental corrections for pressure and salinity, and GLP-compliant data management. The combination of wide measurement range and multiple correction factors makes it suitable for demanding research applications requiring high measurement accuracy.
Practical Tips
Prepare air-saturated water by vigorously aerating distilled water for several minutes, then allowing it to equilibrate at measurement temperature.
Why: Proper saturation ensures accurate 100% oxygen reference point for calibration.
Store the dissolved oxygen probe in appropriate storage solution when not in use and replace the membrane when response becomes sluggish or calibration drift occurs.
Why: Proper probe maintenance extends sensor life and maintains measurement accuracy over time.
Allow sufficient equilibration time for readings to stabilize, especially in samples with low oxygen content or when temperature changes significantly.
Why: Dissolved oxygen sensors require time to reach equilibrium with the sample, particularly at low concentrations.
If readings appear erratic, check for air bubbles under the membrane and ensure adequate sample stirring to maintain oxygen transport to the sensor.
Why: Air bubbles and stagnant conditions can cause measurement artifacts and poor reproducibility.
Record local barometric pressure and sample temperature for each measurement session, especially when working at varying altitudes or during weather changes.
Why: Environmental conditions significantly affect dissolved oxygen saturation calculations and measurement accuracy.
When preparing zero oxygen solutions using sodium sulfite, work in well-ventilated area and follow chemical safety protocols for reagent handling.
Why: Chemical reagents used in calibration solutions require appropriate handling to ensure laboratory safety.
Validate measurements periodically using independent standards or cross-check with alternative dissolved oxygen measurement methods.
Why: Independent validation confirms measurement accuracy and identifies potential systematic errors in the measurement system.
Apply appropriate salinity corrections for samples containing significant dissolved solids, using established conversion factors for your specific sample matrix.
Why: Salinity affects oxygen solubility and uncorrected readings will overestimate dissolved oxygen concentration in saline samples.
Setup Guide
What’s in the Box
- DO400F dissolved oxygen meter (main unit)
- Dissolved oxygen probe with membrane (typical)
- Temperature probe (typical)
- Calibration solutions (typical)
- Power adapter (typical)
- USB cable for data transfer (typical)
- User manual and calibration certificate (typical)
- Probe storage solution (typical)
Warranty
ConductScience provides a standard one-year manufacturer warranty covering defects in materials and workmanship. Technical support includes calibration guidance, troubleshooting assistance, and replacement sensor recommendations for optimal measurement performance.
Compliance
What calibration standards are required for routine use?
The DO400F calibrates using air-saturated water (100% oxygen saturation) and optionally zero oxygen solution prepared with sodium sulfite or nitrogen sparging. Air-saturated water is the most convenient standard as it represents known oxygen concentration at measured temperature and pressure.
How does salinity correction affect measurement accuracy?
Manual salinity factor correction adjusts for reduced oxygen solubility in solutions with high ionic strength. This correction is essential for seawater, brines, or culture media with significant salt content, as uncorrected readings would overestimate actual dissolved oxygen concentration.
What sample volume and stirring requirements apply?
Adequate sample volume should completely immerse the probe sensor and temperature element. Gentle stirring or sample circulation is typically required to ensure representative measurement and prevent oxygen depletion in the immediate sensor vicinity during measurement.
How frequently should the dissolved oxygen probe be replaced?
Probe replacement depends on usage frequency and sample matrix. Membrane replacement is typically needed every 1-3 months with regular use, while electrode replacement may be required annually. Monitor calibration stability and response time as indicators of sensor condition.
What temperature range limitations affect measurement accuracy?
Consult product datasheet for specific temperature range specifications. Temperature compensation is critical as oxygen solubility changes approximately 1.5% per degree Celsius, making accurate temperature measurement essential for reliable dissolved oxygen readings.
How does barometric pressure compensation function?
The meter compensates for atmospheric pressure variations that affect oxygen saturation levels. Local barometric pressure input may be required for highest accuracy, particularly at varying altitudes or during significant weather changes.
What data export formats are supported?
The GLP-compliant data management system stores measurements with time stamps and calibration information. Consult product datasheet for specific data export formats and connectivity options available for integration with laboratory information systems.
How does this compare to optical dissolved oxygen sensors?
Electrochemical sensors like the DO400F typically offer lower initial cost and proven reliability, while optical sensors provide no membrane replacement requirements and resistance to interference from hydrogen sulfide. Consider sample matrix and maintenance preferences when selecting between technologies.