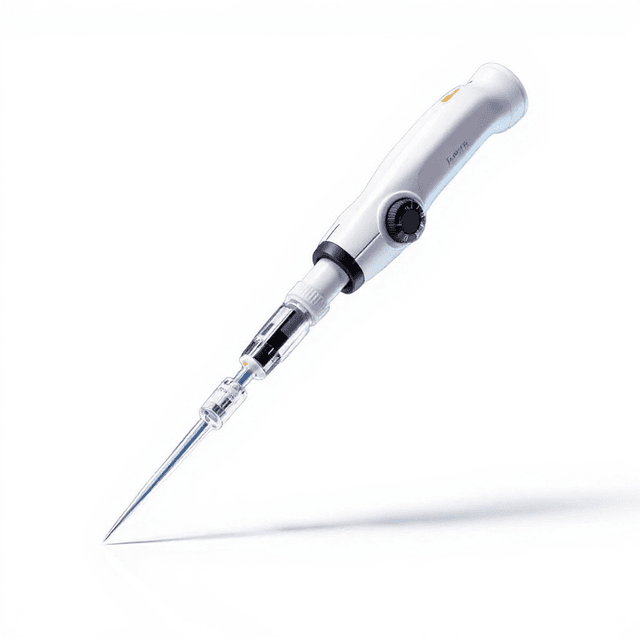
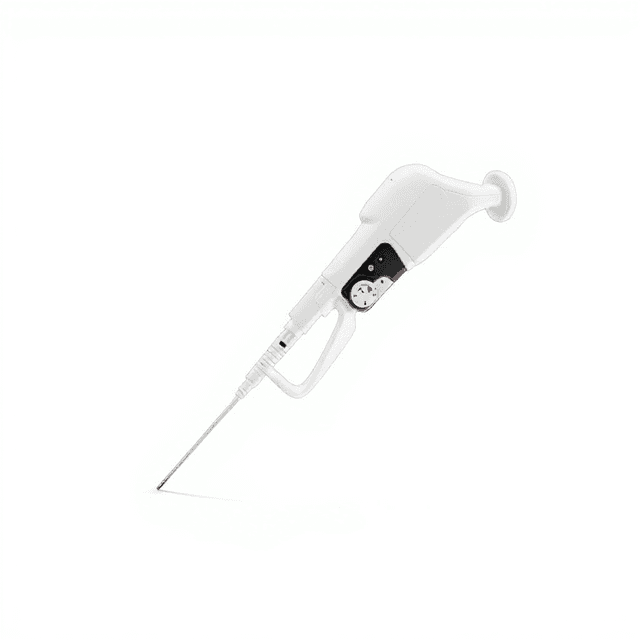
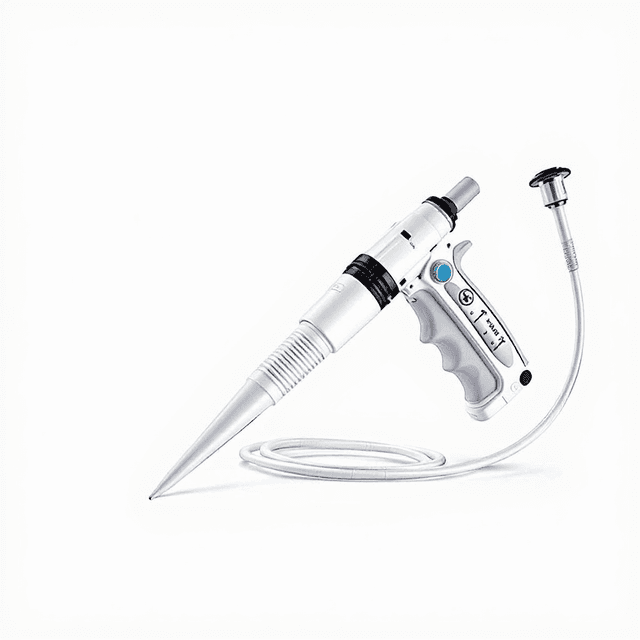
Micro Pipette(Disinfection of Half)
Precision micropipette with increment volume adjustment and documented repeatability specifications for controlled liquid handling in laboratory applications.

Louise Corscadden, PhD
Neuroscience · ConductScience
Ask Louise about Micro Pipette(Disinfection of Half) fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
The Micro Pipette (Disinfection of Half) is a precision liquid handling instrument designed for controlled dispensing and aspiration of small volumes in laboratory applications. This micropipette features increment volume adjustment capabilities with documented capacity tolerance and test repeatability specifications, making it suitable for protocols requiring precise liquid measurement and transfer.
The product incorporates disinfection considerations in its design, addressing laboratory safety protocols for handling potentially contaminated samples. The micropipette's construction allows for reliable performance across repeated dispensing cycles, with tolerance and repeatability parameters that support quantitative experimental workflows.
How It Works
The micropipette operates on an air displacement principle where a piston mechanism creates controlled positive and negative pressure to aspirate and dispense precise liquid volumes. The increment volume adjustment system allows for reproducible setting of specific volumes within the pipette's operating range, with mechanical stops ensuring consistent positioning.
The disinfection design features enable effective decontamination between samples while maintaining measurement accuracy. The internal mechanisms are engineered to minimize carryover and maintain the specified capacity tolerance across repeated use cycles, with the test repeatability parameter indicating the instrument's precision under standardized operating conditions.
Features & Benefits
Automation Level
- manual
Capacity Tolerance(%)
- Test Repeatability(%)
Brand
- ConductScience
Research Domain
- Analytical Chemistry
- Cell Biology
- Clinical Diagnostics
- Materials Science
- Microbiology
- Pharmaceutical QC
Capacity
- Increment
Weight
- 4.41 kg
Dimensions
- L: 42.0 mm
- W: 43.6 mm
- H: 38.0 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Volume Adjustment System | Increment-based adjustment mechanism | Basic models often use continuous adjustment without increment stops | Increment system provides repeatable volume settings and reduces operator variability between measurements |
| Capacity Documentation | Documented capacity tolerance specifications | Entry-level pipettes may lack detailed tolerance documentation | Specified tolerance values support method validation and regulatory compliance requirements |
| Repeatability Testing | Test repeatability specifications provided | Standard models often lack quantified repeatability data | Documented repeatability enables accurate uncertainty calculations in analytical protocols |
| Disinfection Design | Disinfection-compatible construction | Conventional pipettes may require specialized cleaning protocols | Designed compatibility streamlines laboratory safety procedures without compromising measurement accuracy |
This micropipette combines increment-based volume adjustment with documented performance specifications and disinfection-compatible design. The quantified tolerance and repeatability parameters provide measurement traceability, while the disinfection features support laboratory safety protocols.
Practical Tips
Verify calibration using gravimetric analysis with distilled water at room temperature, weighing multiple dispensations to calculate accuracy and precision.
Why: Gravimetric verification provides the most accurate assessment of volume delivery performance.
Inspect and replace tip cone seals when dispensing accuracy degrades or when visible wear is observed.
Why: Worn seals cause air leakage that compromises volume accuracy and repeatability.
Pre-wet tips with the working solution before critical measurements, especially with viscous or volatile liquids.
Why: Pre-wetting eliminates surface tension effects that can cause volume delivery errors in the first few dispensations.
Allow disinfectant solutions to contact all internal surfaces for the specified contact time, then thoroughly rinse with sterile water.
Why: Complete disinfection requires adequate contact time and removal of residual chemicals that could affect subsequent measurements.
If volume delivery becomes inconsistent, check for air bubbles in the tip and ensure complete tip attachment to the cone.
Why: Air leaks are the most common cause of volume delivery problems and are often easily resolved by proper tip seating.
Document pipette serial number and calibration date in experimental records, especially for quantitative studies requiring measurement traceability.
Why: Instrument identification supports data integrity requirements and enables investigation of any measurement discrepancies.
Store the micropipette in a vertical position with the tip ejector engaged to prevent contamination and mechanical damage.
Why: Vertical storage prevents liquid backflow into the piston chamber and protects the volume adjustment mechanism.
Setup Guide
What’s in the Box
- Micropipette main unit (typical)
- User manual and operation guide (typical)
- Calibration certificate (typical)
- Tip fitting adapter (typical)
Warranty
ConductScience provides a one-year manufacturer warranty covering defects in materials and workmanship, with technical support for calibration and maintenance guidance.
Compliance
References
Background reading relevant to this product:
What volume range does the increment adjustment system cover?
The specific volume range and increment settings are detailed in the product datasheet. The increment system allows for precise volume selection within the pipette's operational capacity.
How should the disinfection protocol be implemented without affecting calibration?
Follow manufacturer guidelines for compatible disinfectants and contact times. Avoid harsh chemicals that may degrade seals or affect the piston mechanism. Verify calibration after disinfection protocols.
What is the recommended calibration frequency for this micropipette?
Calibration intervals depend on usage frequency and accuracy requirements. Monthly verification is typical for routine use, with more frequent checks for critical quantitative work or after disinfection cycles.
Can this pipette handle viscous solutions or organic solvents?
Compatibility depends on the specific chemical properties and pipette materials. Consult the chemical compatibility chart in the product documentation before use with non-aqueous solutions.
How does the capacity tolerance specification affect experimental precision?
The capacity tolerance defines the maximum acceptable deviation from the set volume, directly impacting measurement uncertainty in quantitative protocols. Factor this into your error analysis calculations.
What maintenance is required to preserve the repeatability specifications?
Regular cleaning of the tip cone, periodic seal inspection, and proper storage in vertical position. Replace worn seals and service the piston mechanism according to the maintenance schedule.
Have a question about this product?
Accessories
Enhance your setup with compatible accessories