Behavioral Tracking for Water Flea
Daphnia magna
Phototaxis, predator response, and aquatic ecotoxicology in Daphnia magna. ConductVision delivers automated tracking and quantitative parameter extraction across the full assay catalog below.
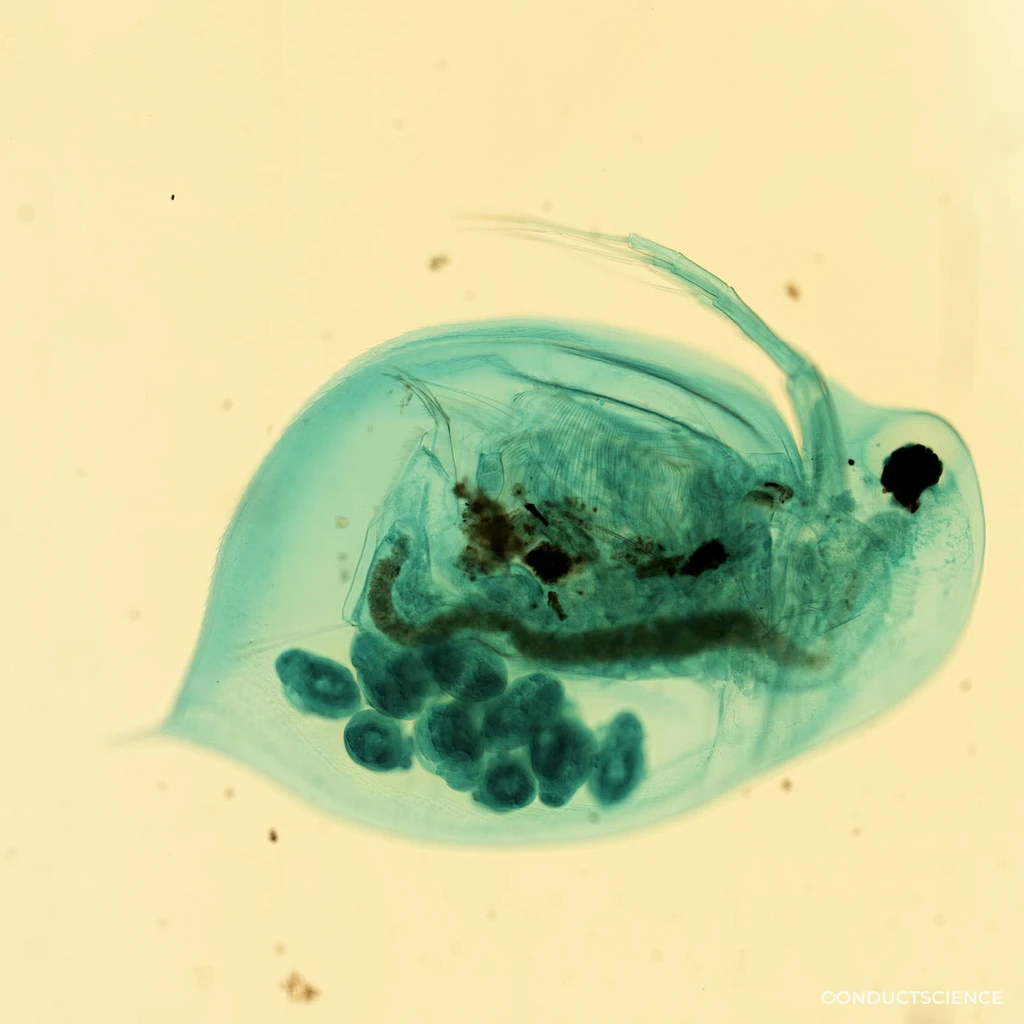
Why Water Flea in Behavioral Research
Daphnia magna is the standard model for aquatic ecotoxicology and a powerful behavioral system for phototaxis, swimming, and predator-induced morphology. Its transparency, short generation time, and clonal reproduction support high-throughput behavioral and developmental studies.
OECD. (2004). Test Guideline 211: Daphnia magna reproduction test.
Tollrian R, Dodson SI. (1999). Inducible defenses in Cladocera: constraints, costs, and multipredator environments. In: Tollrian & Harvell (eds). The Ecology and Evolution of Inducible Defenses, pp 177-202.

What We Measure in Water Flea
Validated assays with quantitative parameter tracking for Daphnia magna.
Daphnia move toward dim light and away from bright illumination, a behavior used in toxicology and circadian research. Vertical and horizontal phototactic indices are core endpoints.
| Parameter | Unit | Description |
|---|---|---|
| Phototactic index | ratio | Light-zone occupancy |
| Swim direction bias | deg | Heading vs light |
| Vertical migration amplitude | mm | Up-down displacement |
| Light threshold | lux | Minimum responsive intensity |
Storz UC, Paul RJ. (1998). Phototaxis in water fleas (Daphnia magna) is differently influenced by visible and UV light. J Comp Physiol A, 183, 709-717.
Kairomones from predatory fish or invertebrates induce morphological defenses (helmets, neck-teeth) and altered swimming. Defense expression and swim depth quantify induced phenotypes.
| Parameter | Unit | Description |
|---|---|---|
| Helmet/neck-teeth size | µm | Morphological defense |
| Mean swim depth | cm | Vertical avoidance |
| Swim speed | mm/s | Activity change |
| Defense induction time | h | Onset after kairomone |
Tollrian R. (1995). Predator-induced morphological defenses: costs, life history shifts, and maternal effects in Daphnia pulex. Ecology, 76(6), 1691-1705.
Spontaneous swimming with characteristic hop-and-sink pattern. Bout frequency, speed, and pause duration are baseline metrics for toxicology assays.
| Parameter | Unit | Description |
|---|---|---|
| Hop frequency | hops/min | Bout rate |
| Mean swim speed | mm/s | Velocity during hop |
| Pause duration | s | Sink phase |
| Total distance | m/h | Cumulative locomotion |
Dodson SI, Hanazato T. (1995). Commentary on effects of anthropogenic and natural organic chemicals on development, swimming behavior, and reproduction of Daphnia, a key member of aquatic ecosystems. Environ Health Perspect, 103 Suppl 4, 7-11. PMID: 7556032
The 24-/48-h immobilization assay is the standard endpoint for chemical toxicity testing in aquatic ecotoxicology. Median effective concentration (EC₅₀) is reported.
| Parameter | Unit | Description |
|---|---|---|
| EC₅₀ at 48h | mg/L | Median effect concentration |
| Immobilization rate | % | Fraction not swimming |
| Mortality | % | Lethal endpoint |
| No-Observed-Effect Concentration | mg/L | NOEC |
OECD. (2004). Test Guideline 202: Daphnia sp. acute immobilisation test.
Heart rate is visible through the transparent carapace and is sensitive to chemical exposure and temperature. Beats-per-minute is a high-throughput cardiotoxicity readout.
| Parameter | Unit | Description |
|---|---|---|
| Heart rate | bpm | Cardiac frequency |
| Heart-rate variability | ms | Beat-to-beat variation |
| Tachycardia threshold | mg/L | Drug-induced HR rise |
| Recovery time | min | Post-exposure return to baseline |
Villegas-Navarro A, et al. (2003). Heart rate of Daphnia magna as toxicity endpoint. Ecotoxicol Environ Saf, 56(2), 269-275. PMID: 12915155
More Behavioral Tests for Water Flea
Feeding Rate
Key Parameters: Algae cleared per individual
Lampert W. (1987). Mem Ist Ital Idrobiol, 45, 143-192.
Reproduction (Brood Size)
Vertical Migration (DVM)
Escape (C-Start) Response
Key Parameters: Turn angle, latency
Pijanowska J, Kowalczewski A. (1997). PMID: 9210081
UV-Induced Behavior
ConductScience Hardware for Water Flea Research
Glass Column Phototaxis Chamber
Vertical phototactic assay
Multi-Well Behavioral Imaging Plate
High-throughput toxicology
Heart-Rate Imaging Microscope
Cardiotoxicity
Predator-Cue Exposure Chamber
Inducible defense induction
Acute Immobilization Test Plate
OECD 202 endpoint
Citations & Further Reading
- OECD. (2004). Test Guideline 211: Daphnia magna reproduction test.
- Tollrian R, Dodson SI. (1999). Inducible defenses in Cladocera: constraints, costs, and multipredator environments. In: Tollrian & Harvell (eds). The Ecology and Evolution of Inducible Defenses, pp 177-202.
- Storz UC, Paul RJ. (1998). Phototaxis in water fleas (Daphnia magna) is differently influenced by visible and UV light. J Comp Physiol A, 183, 709-717.
- Tollrian R. (1995). Predator-induced morphological defenses: costs, life history shifts, and maternal effects in Daphnia pulex. Ecology, 76(6), 1691-1705.
- Dodson SI, Hanazato T. (1995). Commentary on effects of anthropogenic and natural organic chemicals on development, swimming behavior, and reproduction of Daphnia, a key member of aquatic ecosystems. Environ Health Perspect, 103 Suppl 4, 7-11. PMID: 7556032
- OECD. (2004). Test Guideline 202: Daphnia sp. acute immobilisation test.
- Villegas-Navarro A, et al. (2003). Heart rate of Daphnia magna as toxicity endpoint. Ecotoxicol Environ Saf, 56(2), 269-275. PMID: 12915155
Other Model Systems
Discuss Your Daphnia Research
Tell us about your models, assays, and experimental goals — we’ll show you how ConductVision fits your workflow.



