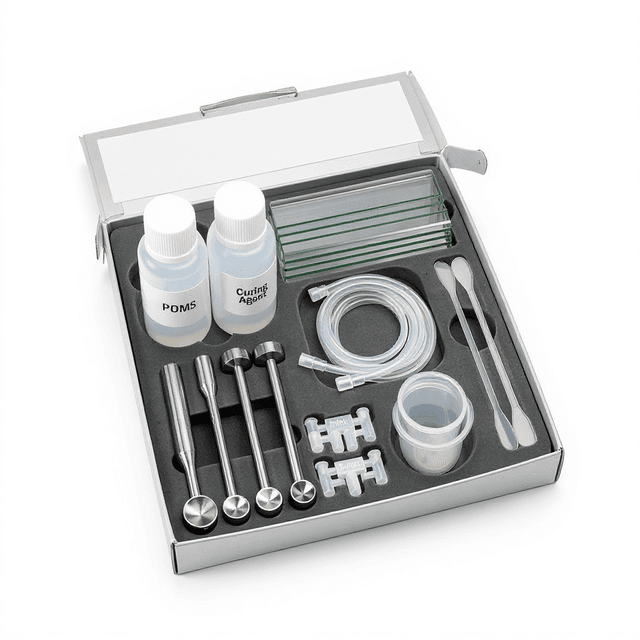
Microfluidic Cell-Cell Interaction Chip
Microfluidic platform for controlled cell-cell interaction studies and co-culture experiments with precise spatial arrangement and fluidic control. Reusable chip — designed for multiple experimental runs. Compatible with standard microfluidic tubing: steel pins (0.7 mm ID / 1.0 mm OD) and silicone tubing (0.8 mm ID / 1.9 mm OD). Available in bulk packs (5‑, 10‑, and 25‑unit) for lab-scale and consumable workflows.

Louise Corscadden, PhD
Neuroscience · ConductScience
Ask Louise about Microfluidic Cell-Cell Interaction Chip fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
The Microfluidic Cell-Cell Interaction Chip provides a controlled platform for studying direct cellular interactions and co-culture experiments in a microscale environment. This device enables researchers to position different cell types in precise spatial arrangements while maintaining controlled fluidic conditions for real-time observation of cell-to-cell communication, signaling pathways, and cooperative behaviors.
The chip's microfluidic architecture allows for controlled nutrient delivery, waste removal, and gradient establishment while cells remain in close proximity for interaction studies. Researchers can monitor cellular responses, migration patterns, and biochemical exchanges between co-cultured populations under controlled experimental conditions. The transparent construction enables direct microscopic observation and imaging of cellular interactions as they occur.
How It Works
The microfluidic cell-cell interaction chip operates through precisely engineered microscale channels and chambers that control cellular positioning and fluid flow. Cells are introduced into designated areas of the chip through separate inlet ports, allowing different cell types to be positioned in specific spatial relationships. The microfluidic channels maintain laminar flow conditions that enable controlled delivery of nutrients, growth factors, or experimental compounds while removing metabolic waste products.
The chip design typically incorporates features such as cell capture areas, perfusion channels, and observation windows that facilitate controlled co-culture conditions. The small scale of the device creates physiologically relevant cell densities and intercellular distances that promote natural cell-to-cell interactions. Transparent materials allow for real-time microscopic observation and time-lapse imaging of cellular behaviors, migration, and morphological changes during interaction studies.
Controlled fluidic gradients can be established across the chip to simulate tissue-like environments or to deliver specific signaling molecules in spatial patterns. This enables researchers to study how cells respond to both direct contact-mediated signaling and paracrine signaling under well-defined experimental conditions.
Features & Benefits
Pack Size
- 5-Pack
- 10-Pack
- 25-Pack
Weight
- 0.03 kg
Dimensions
- L: 25.0 mm
- W: 15.0 mm
- H: 3.0 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Spatial Control | Precise microfluidic channels for controlled cell positioning | Basic well-based systems offer limited spatial organization | Enables study of position-dependent cellular interactions and gradient responses |
| Real-time Observation | Transparent design for continuous microscopic monitoring | Traditional co-cultures require sample destruction for analysis | Allows tracking of interaction dynamics and temporal changes in cell behavior |
| Fluidic Control | Integrated perfusion system with controlled flow | Static culture conditions without flow control | Maintains physiological conditions and enables dynamic experimental manipulations |
| Reagent Consumption | Microscale volumes reduce reagent requirements | Larger culture vessels require substantial media volumes | Reduces experimental costs and enables use of expensive signaling molecules or inhibitors |
This microfluidic chip provides researchers with precise spatial and temporal control over cell-cell interaction studies through integrated perfusion and real-time observation capabilities. The compact design reduces reagent consumption while enabling physiologically relevant co-culture conditions.
Practical Tips
Pre-condition the chip with cell culture medium for 30-60 minutes before cell loading to equilibrate surface properties and remove any residual manufacturing materials.
Why: Conditioning improves cell attachment and reduces experimental variability from surface effects.
Clean chips immediately after use with appropriate detergent solution followed by extensive rinsing to prevent protein buildup in microchannels.
Why: Prompt cleaning prevents channel blockage and maintains consistent flow characteristics for subsequent experiments.
Verify flow rates using dye solutions and measure actual perfusion velocities before each experiment to ensure consistent experimental conditions.
Why: Flow rate calibration ensures reproducible shear conditions and nutrient delivery rates across experiments.
Use multiple imaging positions across the chip to account for potential spatial variations in cell behavior or experimental conditions.
Why: Multiple sampling positions improve statistical power and account for any chip-to-chip or position-dependent variations.
If cells detach during perfusion, reduce flow rates and check surface coating effectiveness; consider using different adhesion molecules for your cell types.
Why: Cell attachment issues often result from excessive shear stress or inadequate surface preparation.
Handle chips carefully to avoid generating sharp fragments if cracked, and dispose of used chips according to biological waste protocols.
Why: Proper handling prevents injury and ensures appropriate disposal of potentially biohazardous materials.
Setup Guide
What’s in the Box
- Microfluidic cell-cell interaction chip
- Inlet/outlet port fittings (typical)
- User guide and protocols (typical)
- Quality control certificate (typical)
Warranty
ConductScience provides a standard 1-year manufacturer warranty covering material defects and workmanship. Technical support is available for experimental protocol optimization and troubleshooting.
Compliance
References
Background reading relevant to this product:
What types of cell-cell interactions can be studied with this chip?
The chip supports studies of direct contact-mediated signaling, paracrine communication, cell migration and invasion, co-culture dynamics, and competitive or cooperative cellular behaviors between different cell types.
How do I maintain sterile conditions during long-term experiments?
Use sterile technique during setup, maintain connections in sterile environment, use filtered media and gases, and consider antibiotic supplementation according to your cell culture protocols.
What microscopy techniques are compatible with this chip?
The transparent design supports bright-field, phase contrast, fluorescence, and confocal microscopy. Time-lapse imaging can track cellular interactions over extended periods.
How do I control chemical gradients across the chip?
Use syringe pumps or pressure-driven flow systems to establish controlled flow rates of different media compositions through separate inlet channels to create stable concentration gradients.
What cell densities work best for interaction studies?
Optimal cell densities depend on cell type and experimental goals. Start with standard culture densities and adjust based on interaction frequency and imaging requirements. Consult product datasheet for recommended seeding protocols.
How do I prevent cell loss during perfusion?
Use low shear flow rates, ensure proper surface coating for adhesion, allow adequate time for cell attachment before initiating flow, and optimize media composition for cell retention.
Can I recover cells after interaction studies?
Cell recovery depends on experimental design and chip configuration. Some protocols allow for cell harvesting through specific outlets or enzymatic detachment procedures.