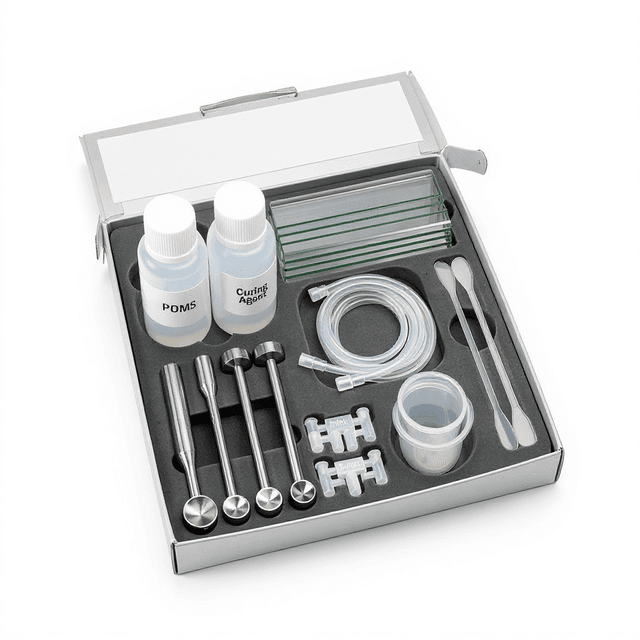
Microfluidic Cell Culture Chip
PDMS microfluidic chip with 200 μm depth channels designed for perfusion-based cell culture applications and long-term cellular behavior studies. Reusable chip — designed for multiple experimental runs. Compatible with standard microfluidic tubing: steel pins (0.7 mm ID / 1.0 mm OD) and silicone tubing (0.8 mm ID / 1.9 mm OD). Available in bulk packs (5‑, 10‑, and 25‑unit) for lab-scale and consumable workflows.

Louise Corscadden, PhD
Neuroscience · ConductScience
Ask Louise about Microfluidic Cell Culture Chip fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
The Microfluidic Cell Culture Chip provides a controlled microenvironment for long-term cell culture studies with integrated perfusion capabilities. This PDMS-based device features precisely engineered channels with 200 μm depth, enabling laminar flow conditions and stable culture gradients essential for cellular behavior analysis. The compact 25 × 15 × 3 mm form factor accommodates standard microscopy setups while minimizing sample volumes and reagent consumption.
The chip supports continuous perfusion protocols for maintaining viable cell cultures over extended periods, facilitating studies of cellular responses to chemical gradients, mechanical stimuli, and temporal variations in culture conditions. The defined channel geometry enables reproducible experimental conditions and quantitative analysis of cell migration, proliferation, and differentiation processes.
How It Works
The microfluidic cell culture chip operates on principles of laminar flow dynamics and controlled mass transfer within microscale channels. The 200 μm channel depth provides sufficient volume for cell attachment and growth while maintaining the high surface-to-volume ratio characteristic of microfluidic systems. This geometry enables rapid equilibration of nutrients and waste products while preventing turbulent mixing that could disrupt cell attachment.
Perfusion flow through the channels maintains stable concentration gradients and removes metabolic waste products, supporting extended culture periods. The laminar flow profile ensures predictable shear stress conditions on cultured cells, with wall shear rates determined by flow rate and channel geometry. The PDMS substrate provides optical transparency for real-time microscopy observation while maintaining biocompatibility and gas permeability essential for cell viability.
Cell seeding occurs through inlet ports, with subsequent perfusion maintaining culture conditions through continuous media exchange. The controlled microenvironment enables temporal studies of cellular processes while minimizing variables associated with traditional culture methods.
Features & Benefits
Pack Size
- 5-Pack
- 10-Pack
- 25-Pack
Weight
- 0.03 kg
Dimensions
- L: 25.0 mm
- W: 15.0 mm
- H: 3.0 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Channel Depth | 200 μm precise depth | Many chips offer 50-100 μm depths | Provides adequate volume for cell attachment while maintaining optimal nutrient transport. |
| Device Size | 25 × 15 mm compact footprint | Larger devices often require specialized microscope stages | Fits standard microscopy setups without additional equipment modifications. |
| Application Focus | Optimized for perfusion-based cell culture | Many devices focus on single-timepoint analysis | Enables extended culture studies and dynamic environmental control. |
| Material Construction | PDMS biocompatible substrate | Some devices use glass or rigid plastics | Provides gas permeability essential for cell viability in closed systems. |
This chip provides a specialized solution for perfusion-based cell culture with optimized channel depth and compact design. The 200 μm channel depth and perfusion capability enable extended culture studies that are challenging with static culture methods.
Practical Tips
Prime all fluidic connections thoroughly before cell seeding to prevent air bubbles that can disrupt flow patterns.
Why: Air bubbles create flow irregularities that affect nutrient transport and cell viability.
Replace tubing and connectors regularly to prevent contamination and maintain consistent flow rates.
Why: Aging tubing can harbor contaminants and develop flow restrictions.
Verify flow rates using microscopic particle tracking or dye injection before each experiment.
Why: Flow rate variations significantly impact shear stress conditions and nutrient transport.
Establish baseline cell behavior in static conditions before transitioning to perfusion protocols.
Why: Baseline data enables identification of flow-dependent cellular responses.
If cells detach unexpectedly, reduce flow rate and verify surface coating quality.
Why: Excessive shear stress or inadequate surface treatment can cause cell loss.
Use sterile technique throughout setup and maintain closed system integrity to prevent contamination.
Why: Microfluidic systems are particularly susceptible to contamination due to small volumes and long culture periods.
Setup Guide
What’s in the Box
- Microfluidic cell culture chip
- User manual with culture protocols (typical)
- Sterile packaging (typical)
Warranty
ConductScience provides a standard 1-year warranty covering manufacturing defects and material quality. Technical support includes protocol guidance and troubleshooting assistance.
Compliance
References
Background reading relevant to this product:
What cell types are compatible with this microfluidic chip?
The chip supports most adherent cell lines and primary cells. Surface treatment selection (fibronectin, collagen, poly-L-lysine) should match specific cell adhesion requirements.
What flow rates are recommended for cell culture applications?
Typical flow rates range from 1-50 μL/hr depending on cell type and experimental objectives. Start with 5-10 μL/hr for most applications and optimize based on cell viability and experimental requirements.
How long can cells be maintained in the chip?
With proper perfusion, most cell types can be cultured for 1-2 weeks. Culture duration depends on cell type, flow conditions, and medium composition.
What microscopy techniques are compatible with this chip?
The chip supports brightfield, phase contrast, fluorescence, and confocal microscopy. PDMS transparency enables high-quality imaging across visible and near-infrared spectra.
How do I prevent bubble formation in the channels?
Prime all tubing and connections before cell seeding. Maintain steady flow rates and avoid air in syringes or reservoirs. Consider degassing media if bubble formation persists.
Can the chip be reused for multiple experiments?
The chip is designed for single-use applications to prevent cross-contamination and ensure reproducible experimental conditions.
What surface treatments are recommended for different cell types?
Use fibronectin for endothelial cells, collagen for hepatocytes, and poly-L-lysine for neural cells. Consult cell culture protocols for specific coating concentrations and incubation times.