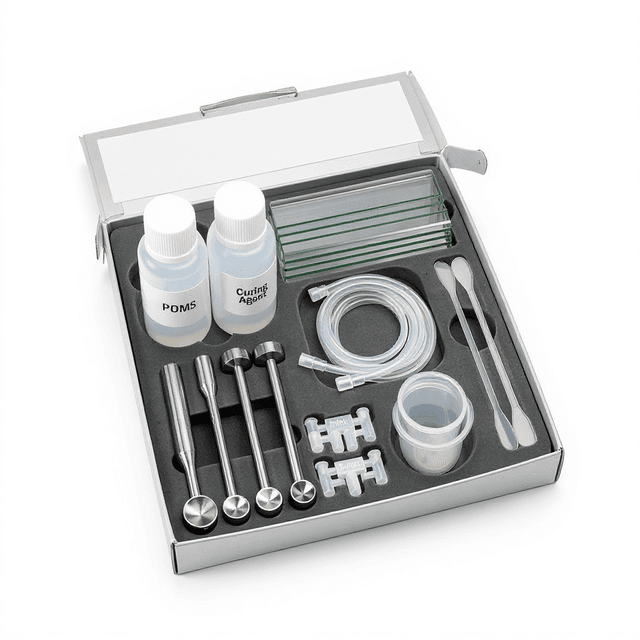
PDMS Cell Sorting Microfluidic Chip
PDMS microfluidic chip with 100-micrometer channels designed for cell sorting and FACS-on-chip applications, providing biocompatible platform for miniaturized cell separation workflows. Reusable chip — designed for multiple experimental runs. Compatible with standard microfluidic tubing: steel pins (0.7 mm ID / 1.0 mm OD) and silicone tubing (0.8 mm ID / 1.9 mm OD). Available in bulk packs (5‑, 10‑, and 25‑unit) for lab-scale and consumable workflows.

Louise Corscadden, PhD
Neuroscience · ConductScience
Ask Louise about PDMS Cell Sorting Microfluidic Chip fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
The PDMS Cell Sorting Microfluidic Chip (WHM-0012) is a polydimethylsiloxane-based microfluidic device engineered for on-chip cell sorting and FACS-on-chip applications. Fabricated with 100-micrometer channel depth, this chip enables precise manipulation and separation of cellular populations in microfluidic environments. The compact 25 × 15 × 3 mm footprint accommodates integration with optical detection systems, microscopy platforms, and automated fluid handling equipment.
PDMS construction provides optical transparency for real-time cell visualization while maintaining biocompatibility for live cell applications. The standardized channel geometry supports consistent fluid dynamics and predictable cell trajectories, enabling reproducible sorting protocols across experimental replicates. This device serves as a platform for developing miniaturized cell analysis workflows that require reduced sample volumes and enhanced spatial control compared to conventional flow cytometry systems.
How It Works
The PDMS Cell Sorting Microfluidic Chip operates on principles of hydrodynamic focusing and laminar flow control within microfabricated channels. Cell suspensions are introduced into the device where they encounter precisely defined flow streams that position cells for analysis or sorting. The 100-micrometer channel depth provides sufficient clearance for most mammalian cell types while maintaining spatial confinement necessary for predictable flow patterns.
Sorting mechanisms depend on the specific implementation but typically involve fluorescence-activated switching, dielectrophoretic forces, or acoustic actuation to direct target cells into collection channels. The laminar flow regime at microscale Reynolds numbers ensures minimal mixing between adjacent flow streams, enabling clean separation of sorted populations. PDMS material properties allow for rapid prototyping of custom channel geometries while providing the optical clarity required for fluorescence detection and cell tracking.
Integration with external detection systems enables real-time analysis of cell properties during transit through the device. The chip can be operated under continuous flow conditions or with stop-flow protocols depending on the specific sorting criteria and downstream analysis requirements.
Features & Benefits
Pack Size
- 5-Pack
- 10-Pack
- 25-Pack
Weight
- 0.02 kg
Dimensions
- L: 25.0 mm
- W: 15.0 mm
- H: 3.0 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Channel Depth | 100 micrometers | Basic devices often use 50-75 micrometer depths | Greater depth accommodation reduces clogging risk with larger cell types and cell aggregates |
| Material Construction | PDMS substrate | Glass or silicon-based alternatives available | PDMS provides superior optical clarity and biocompatibility while enabling rapid prototyping of custom designs |
| Device Footprint | 25 × 15 mm compact design | Standard microfluidic chips vary widely in size | Compact dimensions enable easy integration with existing microscopy setups and automated platforms |
| Application Focus | Cell sorting and FACS-on-chip | General-purpose microfluidic devices may lack sorting optimization | Purpose-built design ensures optimal channel geometry and flow characteristics for cell separation applications |
This PDMS microfluidic chip provides 100-micrometer channel depth in a compact 25×15 mm format specifically optimized for cell sorting applications. The PDMS construction offers excellent optical properties for fluorescence detection while maintaining biocompatibility for live cell work.
Practical Tips
Verify channel dimensions and flow characteristics using fluorescent microspheres before cell experiments.
Why: Pre-validation ensures proper device performance and helps optimize flow conditions for specific cell types.
Store chips in sterile packaging and avoid exposure to organic solvents that may cause PDMS swelling.
Why: Proper storage maintains channel integrity and prevents dimensional changes that affect sorting performance.
Filter cell suspensions through 40-micrometer mesh before loading to remove debris and cell clumps.
Why: Pre-filtration prevents channel clogging and ensures consistent cell flow through sorting regions.
If cells stick to channel walls, adjust buffer composition or add blocking agents like BSA or Pluronic F-127.
Why: Surface treatments reduce non-specific cell adhesion that can compromise sorting efficiency and cell viability.
Record flow rates, cell concentrations, and environmental conditions for each experiment.
Why: Documentation enables reproducible results and helps optimize protocols across different experimental conditions.
Use appropriate biosafety containment when working with pathogenic cells or toxic reagents.
Why: Microfluidic devices may generate aerosols during operation, requiring proper safety precautions for hazardous materials.
Setup Guide
What’s in the Box
- PDMS Cell Sorting Microfluidic Chip
- User manual with protocol recommendations (typical)
- Certificate of fabrication specifications (typical)
Warranty
ConductScience provides standard warranty coverage for microfluidic devices against manufacturing defects. Technical support includes protocol guidance and troubleshooting assistance for chip-based cell sorting applications.
Compliance
References
Background reading relevant to this product:
What cell sizes are compatible with the 100-micrometer channel depth?
The 100-micrometer channel depth accommodates most mammalian cell types ranging from 10-30 micrometers in diameter, including primary cells, cancer cell lines, and stem cells, while providing sufficient clearance to prevent clogging.
How is the chip sterilized before cell sorting experiments?
PDMS chips can be sterilized using UV irradiation, gamma sterilization, or ethylene oxide treatment. For immediate use, sterile filtration of all solutions and aseptic handling techniques are typically sufficient.
What flow rates are recommended for optimal cell sorting performance?
Optimal flow rates depend on cell type and sorting criteria but typically range from 1-100 μL/min for sample flow, with sheath flows 2-10 times higher to achieve proper hydrodynamic focusing. Consult product datasheet for specific recommendations.
Can the chip be reused for multiple sorting experiments?
While PDMS can withstand multiple uses, single-use operation is recommended to prevent cross-contamination and ensure consistent performance, especially when working with different cell types or valuable samples.
What microscopy setup is required for fluorescence detection?
The chip requires inverted fluorescence microscope with appropriate filter sets, high-speed camera for real-time imaging, and optionally laser excitation sources depending on fluorophores used in cell labeling protocols.
How does sorting efficiency compare to conventional flow cytometry?
Microfluidic sorting typically achieves lower throughput (hundreds to thousands of cells per second) compared to conventional FACS but offers advantages in sample volume reduction, spatial precision, and integration with downstream analysis.
What are the main sources of cell damage during chip-based sorting?
Potential damage sources include shear stress from high flow rates, osmotic stress from buffer composition changes, and mechanical stress at channel constrictions. Optimization of flow conditions and buffer formulations minimizes cell viability loss.
Is the PDMS microfluidic chip reusable?
Yes. The PDMS chip is reusable.
What tubing is used to connect to the chip inlets and outlets?
The inlet/outlet holes have a diameter of 1 mm. The chip ships with a steel pin (inner diameter 0.7 mm, outer diameter 1.0 mm) and a silicone tube (inner diameter 0.8 mm, outer diameter 1.9 mm) for connection.