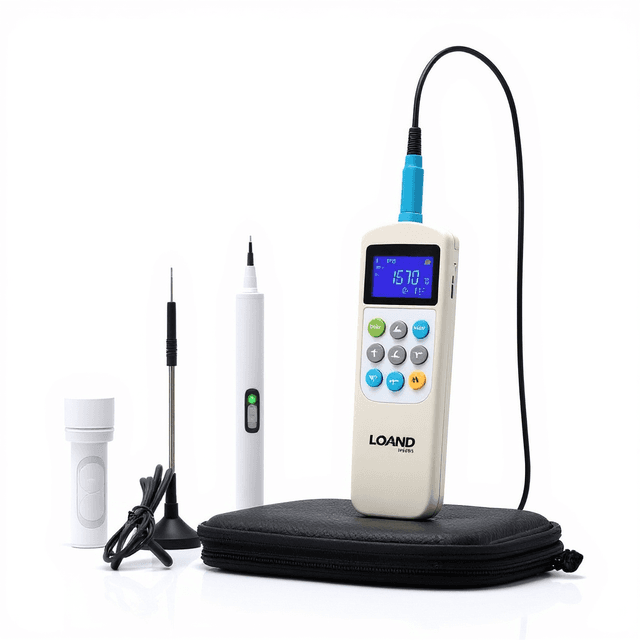
Portable pH, ORP, and Temperature Analyzer
Portable electrochemical analyzer providing simultaneous pH (0-14), ORP (-1999 to +1999 mV), and temperature (0-80°C) measurement with laboratory-grade accuracy for field water quality assessment.
| pH Resolution | 0.1 pH |
| pH Accuracy | +/-0.01 pH |
| ORP Range | -1999 to 1999 mV |
| ORP Accuracy | +/-0.1% F.S. |
| pH Range | 0-14 pH |
| Temperature Accuracy | +/-0.5 C |
The Portable pH, ORP, and Temperature Analyzer provides simultaneous electrochemical measurement of pH (0-14), oxidation-reduction potential (-1999 to +1999 mV), and temperature (0-80°C) in aqueous solutions. This handheld instrument utilizes combination glass electrodes for pH determination and platinum electrodes for ORP measurement, delivering laboratory-grade accuracy in field-portable format.
The analyzer features a backlit LCD display for clear readout in variable lighting conditions and operates on standard AAA batteries for extended field deployment. With pH accuracy of ±0.01 units and ORP accuracy of ±0.1% full scale, the instrument supports quantitative water quality assessment protocols across research and industrial applications.
How It Works
pH measurement employs a glass membrane electrode system containing internal reference solution separated from the sample by a pH-selective glass membrane. When immersed in solution, hydrogen ions interact with the glass surface, generating a potential difference proportional to pH according to the Nernst equation. The instrument measures this millivolt signal and converts it to pH units using temperature compensation algorithms.
ORP measurement utilizes a platinum working electrode paired with a reference electrode to measure the solution's oxidation-reduction potential. The platinum surface responds to electron transfer reactions in the sample, generating a voltage signal that reflects the solution's tendency to gain or lose electrons. Temperature measurement employs integrated thermistors providing automatic temperature compensation for both pH and ORP readings.
The backlit LCD displays simultaneous readings from all three parameters, with internal microprocessor handling signal conditioning, temperature compensation, and digital conversion of the electrochemical signals.
Features & Benefits
pH Resolution
- 0.1 pH
pH Accuracy
- +/-0.01 pH
ORP Range
- -1999 to 1999 mV
ORP Accuracy
- +/-0.1% F.S.
Research Domain
- Analytical Chemistry
- Environmental Monitoring
- Food Science
- Industrial Hygiene
- Microbiology
- Pharmaceutical QC
pH Range
- 0-14 pH
Temperature Accuracy
- +/-0.5 C
Automation Level
- manual
Brand
- ConductScience
Temperature Range
- 0-80 C
Display Type
- Backlit LCD
Power/Voltage
- 3x AAA batteries
Weight
- 0.27 kg
Dimensions
- L: 165.0 mm
- W: 75.0 mm
- H: 51.0 mm
Comparison Guide
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| pH Accuracy | ±0.01 pH | Entry-level models often offer ±0.02 pH or lower precision | Higher accuracy enables more precise pH control in critical research applications and quality control procedures. |
| ORP Measurement Range | -1999 to +1999 mV | Basic analyzers may offer narrower ranges or lack ORP capability | Full-scale ORP measurement captures both strongly oxidizing and reducing conditions in diverse sample types. |
| Multi-Parameter Display | Simultaneous pH, ORP, and temperature display | Single-parameter instruments require separate measurements | Reduces measurement time and ensures all parameters are recorded under identical sample conditions. |
| Temperature Compensation | Automatic compensation for 0-80°C range | Manual compensation or limited temperature ranges in basic models | Maintains measurement accuracy across varying sample temperatures without manual intervention. |
| Power Supply | 3x AAA batteries | Some models require proprietary batteries or AC power | Standard batteries ensure continuous field operation without specialized charging equipment. |
| Display Technology | Backlit LCD | Non-backlit displays limit visibility in poor lighting | Clear readout capability in variable field lighting conditions improves measurement reliability. |
The analyzer provides laboratory-grade pH accuracy (±0.01) combined with comprehensive ORP measurement and automatic temperature compensation in a portable format. The multi-parameter capability and standard battery operation support efficient field water quality assessment protocols.
Practical Tips
Use fresh buffer solutions for calibration and ensure buffer temperatures match sample temperatures when possible.
Why: Buffer solution aging and temperature differences can introduce systematic errors in calibration.
Store pH electrodes in appropriate storage solution (not distilled water) and keep ORP electrode surfaces clean and uncontaminated.
Why: Proper storage maintains electrode sensitivity and extends operational lifetime.
Allow sufficient time for electrode equilibration in each sample before recording readings, especially for ORP measurements.
Why: Redox equilibrium can be slow to establish, and premature readings may not represent true sample conditions.
If pH readings drift or respond slowly, clean electrodes thoroughly and check for glass membrane damage or reference junction blockage.
Why: Electrode contamination or damage are common causes of poor response and measurement instability.
Record sample temperature along with pH and ORP values, even with automatic compensation enabled.
Why: Temperature data provides context for interpreting measurements and troubleshooting any compensation issues.
Rinse electrodes with distilled water between samples to prevent cross-contamination, especially when measuring samples with extreme pH values.
Why: Sample carryover can affect subsequent measurements and potentially damage electrodes through chemical incompatibilities.
Verify ORP calibration using multiple standard solutions spanning the expected measurement range for your application.
Why: ORP electrode response can be non-linear across wide potential ranges, requiring multi-point verification for accuracy.
Minimize exposure of samples to atmospheric oxygen when measuring ORP in anaerobic or low-redox samples.
Why: Atmospheric oxygen can rapidly alter the redox potential of oxygen-sensitive samples, leading to inaccurate measurements.
Setup Guide
What’s in the Box
- Portable pH/ORP/Temperature Analyzer (main unit)
- pH combination electrode (typical)
- ORP platinum electrode (typical)
- Temperature sensor probe (typical)
- Calibration buffer solutions (typical)
- Carrying case (typical)
- User manual and calibration certificate (typical)
- 3x AAA batteries (typical)
Warranty
ConductScience provides a standard one-year manufacturer warranty covering defects in materials and workmanship, with technical support available for calibration procedures and operational guidance.
Compliance
References
Background reading relevant to this product:
What calibration frequency is recommended for maintaining measurement accuracy?
Daily calibration is recommended for critical applications, with pH calibration using 3-point buffer solutions (pH 4, 7, 10) and ORP calibration using standard redox solutions. Weekly calibration may suffice for routine monitoring applications.
How does temperature affect pH and ORP measurements?
The analyzer provides automatic temperature compensation for pH measurements following Nernst equation principles. ORP measurements are also temperature-compensated, though the effect is generally smaller than for pH. Manual temperature coefficient adjustment may be available for specific sample types.
What electrode maintenance is required for optimal performance?
pH electrodes require storage in pH 4 buffer or electrode storage solution, regular cleaning to remove sample residue, and periodic replacement when response becomes sluggish. ORP electrodes need cleaning with appropriate solvents and occasional polishing of the platinum surface.
Can the analyzer measure pH in non-aqueous or high-ionic-strength solutions?
Standard glass electrodes are optimized for aqueous solutions with moderate ionic strength. Non-aqueous measurements may require specialized electrodes, and high-ionic-strength solutions may affect accuracy. Consult product datasheet for specific sample matrix limitations.
What is the typical response time for stable readings?
pH readings typically stabilize within 30-60 seconds in well-buffered solutions, though poorly buffered samples may require longer. ORP measurements may need 2-5 minutes to reach equilibrium, particularly in samples with complex redox chemistry.
How does this compare to laboratory bench-top pH meters?
The portable analyzer offers similar accuracy (±0.01 pH) to bench-top instruments while providing field portability. Laboratory meters may offer additional features like data logging, multiple electrode inputs, or specialized measurement modes not available in portable formats.
What data output options are available?
Consult product datasheet for specific data output capabilities, which may include digital interfaces, data logging functions, or connectivity options for transferring measurements to external devices or software.
Have a question about this product?
Accessories
Enhance your setup with compatible accessories