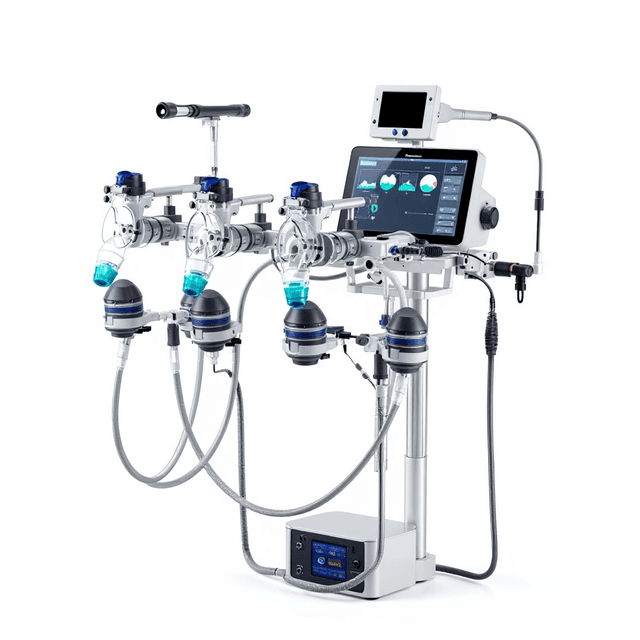
Anesthesia Induction Chamber
The Anesthesia Induction Chamber is used to confine the rodent in a closed space during anesthesia procedures. The transparent material helps in visualizing the physical state of the subject; thus preventing accidental overdosing. An anesthesia in...
| inlet_air_flow_range | 0.1-4L/min |
| bellows_length | 1.2m |
| gas_filter_canister_model | R510-31S |
| anesthesia_duration | 2 to 5 minutes |
| wait_time_before_opening | 10 to 15 seconds |
| compatible_gases | Almost all non-explosive gas mixtures |
Specifications
Anesthesia Induction Chambers
| Model | Species | Dimensions [L x W x H] |
|---|---|---|
| RWD-AICM-V100 | Mouse | 15 x 10 x 10 cm |
| RWD-AICR-V101 | Rat | 24 x 12 x 18 cm |
| RWD-AICRC-V102 | Rabbit/Cat | 40 x 18.5 x 25 cm |
Each model contains 1 plexiglass box, 1.2m bellows and 1 gas filter canister (R510-31S)
Low Stress Anesthesia Induction Chamber
| Model | Species | Dimensions [L x W x H] |
|---|---|---|
| RWD-AICLS-V105 | Mice and Rats | 29 x 15 x 17 cm |
| RWD-AICLS-V106 | Mice | 16 x 10 x 11 cm |
The pipeline connecting the Gas Evacuation. Apparatus is already included
Low stress Anesthesia Induction chamber
Rodents lack cone cells that sense red light and are insensitive to red light.The red transparent appearance is convenient for the experimenter to observe the stateof the animal while reducing the stress response of the animal and reducing the impacton the biological rhythm of the animal. Comply with animal welfare.
Anesthesia Induction Chambers
ConductScience’s Anesthesia Induction Chamber is a state-of-the-art apparatus contrived from supreme quality acrylic. It is used to confine subjects during an induction procedure. The box is provided with two adjacent orifices; an inlet for the entry of the fresh anesthetizing gas and an outlet for scavenging the waste gas material. The transparent material helps in visualizing the physical state of the subject; thus preventing accidental overdosing. The Anesthesia Induction Chamber can be controlled by a flowmeter with inlet air ranging from 0.1-4L/min.
The subject(s) should be carefully placed into the box, and the supply of fresh anesthetizing should be initiated and maintained for approximately 2 to 5 minutes. After the subject is fully anesthetized, the anesthesia supply should be discontinued.
Critical: Do not open the anesthesia box immediately, wait for about 10 to 15 seconds so that oxygen in the box neutralizes the gas concentration.
Finally, the chamber should be opened slightly, just enough to remove the anesthetized subject from the box, and closed immediately.
Caution: Do not wide open the chamber, just enough so that a hand can be introduced for the removal of the subject.
By keeping the opening far away from the experimenter and limiting the time that the box is open, the risk of exposure to the experimenter will be diminished.
Optimal Benefits Achieved Through Our Exceptional Design
- Our design is preferred over the conventional mask and circuits because it is less time-consuming and it can hold and anesthetize more than one subject simultaneously.
- Our special design provides extra protection to the experimenter by keeping the opening far away from the experimenter.Our device is compatible with almost all (non-explosive) gas mixtures. However, it should not be used with liquid organic solvents.
- Our scavenging tube efficiently removes the waste gas materials.
A fundamental component of surgical procedures in animal research is the usage of anesthetics. Anesthesia is an important tool to restrain animals during procedures that may either cause excessive stress to the animal or expose the researcher to unavoidable hazards, as well as post-surgery pain management. The surgical methods that are sure to cause pain or discomfort in animals must, therefore, be performed under general anesthesia (“Anesthesia and Analgesia in Laboratory Animals,” 2010). Anesthesia may be inhaled or injected depending on factors such as type and duration of surgery and animal species.
The process of putting animals under inhalant anesthesia is made easier with the use of an anesthesia box that confines subjects during the anesthesia induction procedure. It is also provided with two separate openings, an inlet for the entry of fresh anesthetizing gas and an outlet for scavenging the waste gas material. To prevent accidental overdosing, the apparatus is made of transparent acrylic for easy observation of the animal
Apparatus and Equipment
The anesthesia box is a state-of-the-art apparatus manufactured from supreme quality acrylic, used to enclose animals during inhalant anesthesia induction procedures. The transparent acrylic material is used for convenient inspection of the animal’s physical state, in order to prevent any possibility of accidental overdose.
Protocol
The anesthesia box must be carefully placed on an even and stable surface. Depending on its size, the anesthesia box may accommodate more than one animal.
Once the animal is carefully placed inside the empty anesthesia box, the supply of fresh anesthetizing gas is then initiated and maintained for approximately 2 to 5 minutes. The supply should then be discontinued once the animal is fully anesthetized. A critical step is to wait for about 10 to 15 seconds after the anesthesia supply is cut off so that the oxygen in the box neutralizes the gas concentration. Once the animal is retrieved through a slight opening in the top lid, the anesthesia box is immediately closed to prevent the unwanted escape of gas. Once the animal is moved out of the chamber, anesthesia may be maintained via other tools, such as the cone device.
Applications
The discovery of general anesthesia during the middle of the 19th century can be considered one of the most important developments in the history of medicine (Werner et al., 2011), and since then, its usefulness has found other important applications, such as animal research.
Much of research in the neurosciences is benefited from the use of experimental animals. A huge aspect of animal research is the employment of surgical procedures, and ethical considerations necessitate anesthesia as a tool for eliminating any unnecessary stress, pain, or discomfort the animal may experience during and after surgery. Today, there are many novel and sophisticated methods for administering different types of anesthesia, but the mechanisms have not changed. The anesthesia box presents a convenient way of employing gas induction procedures, with such substances as isoflurane, sevoflurane, and other inhalant agents, that, through inlet and outlet openings of gas and a transparent, sealed design, ensures the safety of everybody involved.
Strengths and Limitations
The anesthesia box’s design is much preferred over the conventional mask and circuit method because it requires less time for the inhalant anesthesia to take effect. Further, the anesthesia box can hold and anesthetize more than one animal simultaneously, depending on its size.
It is a reality that exposure to waste anesthetic gas is a serious occupational hazard (“Anesthesia and Analgesia in Research Animals,” 2012). The experimenter’s safety is, therefore, another concern that the anesthetic box’s design succeeds in addressing. The anesthesia box’s special design provides extra protection to the experimenter by keeping the opening on the farther side to avoid exposure. Additionally, the anesthesia box is compatible with most non-explosive gas mixtures. The outlet scavenging tube is also an efficient addition to the anesthesia box’s design, effectively removing toxic waste gas materials.
Summary
- An anesthesia box is a tool used for confining animal subjects in a closed space during anesthesia induction procedures.
- To prevent accidental overdosing, the apparatus is made of transparent acrylic for easy observation of the physical state of the animal.
- The anesthesia box ensures the safety of experimenters from accidental exposure to waste anesthetic gas.
References
Anesthesia and Analgesia in Laboratory Animals. (2010, November 08). Retrieved from Penn State Animal Research Program, https://www.research.psu.edu/arp/anesthesia.html
Anesthesia and Analgesia in Research Animals. (2012, December 01). Retrieved from https://lar.indiana.edu/doc/Anesthesia_and_Analgesia_in_Research_Animals.pdf
Werner, D. F., Swihart, A., Rau, V., Jia, F., Borghese, C. M., McCracken, M. L., ... & Eger, E. I. (2011). Inhaled anesthetic responses of recombinant receptors and knockin mice harboring α2 (S270H/L277A) GABAA receptor subunits that are resistant to isoflurane. Journal of Pharmacology and Experimental Therapeutics, 336(1), 134-144.[/vc_column_text][vc_column_text]Quan Ren, Mian Peng, Yuanlin Dong, Yiying Zhang, Ming Chen, Ning Yin, Edward R. Marcantonio, Zhongcong Xie. (2015). Surgery plus anesthesia induces loss of attention in mice. Front Cell Neurosci, 9: 346
Amy Miller, Gemma Kitson, Benjamin Skalkoyannis, Matthew Leach. (2015). The effect of isoflurane anaesthesia and buprenorphine on the mouse grimace scale and behaviour in CBA and DBA/2 mice. Appl Anim Behav Sci, 172: 58–62
Lundt A, Wormuth C, Siwek ME, Muller R, Ehninger D, Henseler C. (2016). EEG Radiotelemetry in Small Laboratory Rodents: A Powerful State-of-the Art Approach in Neuropsychiatric, Neurodegenerative, and Epilepsy Research. Neural Plast, 8213878
F. Werner, A. Swihart, V. Rau, F. Jia, C. M. Borghese, M. L. McCracken, S. Iyer, M. S. Fanselow, I. Oh, J. M. Sonner, E. I. Eger, N. L. Harrison, R. A. Harris, G. E. Homanics. (2011). Inhaled Anesthetic Responses of Recombinant Receptors and Knockin Mice Harboring α2(S270H/L277A) GABAA Receptor Subunits That Are Resistant to Isoflurane. Journal of Pharmacology and Experimental Therapeutics January, 336 (1) 134-144
Michael A. Makara, Ky V. Hoang, Latha P. Ganesan, Elliot D. Crouser, John S. Gunn, Joanne Turner, Larry S. Schlesinger, Peter J. Mohler, Murugesan V.S. Rajaram. (2016). Cardiac Electrical and Structural Changes During Bacterial Infection: An Instructive Model to Study Cardiac Dysfunction in Sepsis. Journal of the American Heart Association, 5:e003820
Manufacturer Source: https://www.rwdstco.com/product-item/anesthesia-induction-chambers-2/
How It Works
Gas anesthesia induction operates through pulmonary uptake of volatile anesthetic agents that cross the blood-brain barrier to depress central nervous system activity. Inhalant agents such as isoflurane activate GABA and glycine receptors while inhibiting NMDA receptors, producing unconsciousness, analgesia, and muscle relaxation. The chamber creates a controlled atmosphere where anesthetic vapor concentration can be precisely managed through the inlet flow system.
The plexiglass chamber confines the animal within the anesthetic atmosphere while allowing visual monitoring of respiratory patterns and response to stimulation. Gas circulation through the 1.2m bellows system ensures uniform anesthetic distribution throughout the chamber volume. The integrated filtration system captures waste gases, preventing environmental contamination and operator exposure.
Red transparent construction in low-stress models exploits rodent photoreceptor physiology, as these species lack red-sensitive cone cells, reducing visual stress during induction while maintaining researcher observation capability. This approach minimizes stress-induced physiological changes that could confound experimental results.
Features & Benefits
inlet_air_flow_range
- 0.1-4L/min
bellows_length
- 1.2m
gas_filter_canister_model
- R510-31S
anesthesia_duration
- 2 to 5 minutes
wait_time_before_opening
- 10 to 15 seconds
compatible_gases
- Almost all non-explosive gas mixtures
incompatible_substances
- Liquid organic solvents
includes_gas_evacuation_pipeline
- Yes
Automation Level
- manual
Material
- Acrylic
- Plexiglass
Color
- Red transparent
Brand
- RWD
Research Domain
- Behavioral Pharmacology
- Cancer Research
- Cardiovascular
- Developmental Biology
- Metabolic Research
- Neuroscience
- Toxicology
Species
- Rabbit
- Mouse
- Rat
Model
- Mouse
- Rat
- Rabbit/Cat
- Mouse Low-Stress (Small)
- Mouse+Rat Low-Stress
Weight
- 8.27 kg
Dimensions
- L: 34.0 mm
- W: 39.0 mm
- H: 33.0 mm
Comparison Guide
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Chamber Volume Options | Three species-specific sizes (1.5L, 5.2L, 18.5L) plus multi-species low-stress model | Many systems offer single-size chambers requiring size adaptation | Optimized chamber volume ensures appropriate anesthetic concentration for each species without oversized chambers that waste anesthetic agents. |
| Stress Reduction Technology | Red transparent construction exploiting rodent photoreceptor insensitivity | Standard clear chambers provide full spectrum light exposure | Reduces stress-induced physiological changes that can confound experimental measurements and improve animal welfare. |
| Flow Rate Control Range | 0.1-4 L/min adjustable inlet flow with precision control | Fixed flow systems often limit protocol flexibility | Accommodates different chamber sizes and anesthetic protocols for consistent induction timing across varied experimental designs. |
| Waste Gas Management | Integrated evacuation pipeline with R510-31S filter canister | Separate scavenging systems require additional setup | Built-in waste gas capture simplifies installation and ensures operator safety without additional equipment purchases. |
| Induction Timeline | 2-5 minute controlled induction with 10-15 second clearance protocol | Variable induction times with less defined clearance procedures | Standardized timing protocols improve experimental reproducibility and reduce variability in anesthetic effects on study outcomes. |
This chamber system combines species-specific sizing, stress reduction technology, and integrated safety features to provide controlled anesthetic induction with enhanced animal welfare considerations. The modular design accommodates multiple research applications while maintaining consistent performance across different experimental protocols.
Practical Tips
Verify anesthetic concentration using a vapor analyzer at different flow rates to establish reproducible induction protocols.
Why: Concentration verification ensures consistent anesthetic delivery and reduces variability between experimental sessions.
Replace the R510-31S gas filter canister when breakthrough odor is detected or after specified usage volume.
Why: Saturated filters reduce scavenging efficiency and may expose personnel to waste anesthetic vapors.
Pre-fill the chamber with anesthetic gas before animal placement to minimize induction time and handling stress.
Why: Pre-saturation reduces the time animals spend in an unfamiliar environment before anesthetic effects begin.
Ensure chamber is completely cleared of anesthetic gases before opening using the specified 10-15 second wait period.
Why: Residual anesthetic vapors can cause personnel exposure and affect the animal's recovery trajectory.
If induction takes longer than 5 minutes, check for chamber leaks and verify vaporizer output concentration.
Why: Extended induction times often indicate inadequate anesthetic delivery rather than animal resistance to anesthesia.
Document anesthetic concentration, flow rate, and induction time for each animal to identify protocol optimization opportunities.
Why: Systematic documentation enables protocol refinement and helps identify sources of experimental variability.
Use the red transparent chamber for studies involving stress-sensitive endpoints such as hormone measurements or cardiovascular parameters.
Why: Reduced stress during induction prevents confounding physiological changes that could affect study outcomes.
Clean chambers with laboratory detergent after each use and inspect acrylic surfaces for stress cracks or chemical damage.
Why: Regular cleaning prevents contamination between animals and surface inspection identifies potential failure points before gas leakage occurs.
Setup Guide
What’s in the Box
- Plexiglass anesthesia chamber (species-specific size)
- 1.2m bellows tubing system
- R510-31S gas filter canister
- Gas evacuation pipeline connections
- Chamber lid with secure closure mechanism
- User manual with setup instructions
Warranty
ConductScience provides a standard one-year manufacturer warranty covering defects in materials and workmanship, with technical support for setup and operational guidance.
Compliance
References
Background reading relevant to this product:
What anesthetic agents are compatible with this chamber system?
The chamber supports almost all non-explosive gas mixtures including isoflurane, sevoflurane, and halothane. Liquid organic solvents are not compatible with the acrylic construction and should be avoided.
How do I determine the appropriate flow rate for my experimental protocol?
Flow rates between 0.1-4 L/min should be adjusted based on chamber volume and desired induction time. Smaller chambers require lower flow rates, while larger animals need higher flows to achieve uniform anesthetic distribution.
What maintenance is required for consistent performance?
Regular cleaning with laboratory detergents, inspection of tubing connections for wear, and replacement of the R510-31S gas filter canister based on usage volume. Check chamber seal integrity monthly to prevent gas leakage.
Can multiple animals be induced simultaneously in the larger chambers?
The low-stress model accommodates multiple mice simultaneously, but individual housing is recommended to prevent stress from social interaction during anesthetic induction. Monitor each animal individually for appropriate response.
How does the red transparent design affect experimental protocols?
Red transparent construction reduces stress-induced physiological changes in rodents while maintaining observation capability. This is particularly important for studies measuring stress hormones or cardiovascular parameters where pre-anesthetic stress could confound results.
What safety measures are required for waste gas management?
Connect the evacuation system to laboratory fume hood or dedicated scavenging unit. Ensure adequate room ventilation and consider personnel monitoring for anesthetic exposure in high-usage environments.
How do I verify proper anesthetic depth before removing animals?
Monitor respiratory rate, response to toe pinch, and righting reflex through the transparent walls. Wait 10-15 seconds after procedure completion before opening chamber to ensure adequate anesthetic clearance.
What are the temperature considerations for chamber operation?
Maintain ambient laboratory temperature as anesthetic vapor pressure varies with temperature. Cold chambers may require longer equilibration times, while excessive heat can increase evaporation rates and alter anesthetic concentrations.
Have a question about this product?
Accessories
Enhance your setup with compatible accessories