The picturesque path of scientific progress is marked by numerous challenges and obstacles. For instance, medical research and clinical trials, in particular, face a wide variety of practical issues. Recruitment, funding, and deadlines are only some of them. Often professionals and sponsors focus on participants, procedures, and outcomes. As a result, researchers may forget about one of the most crucial aspects: Good documentation. However, professionals worldwide agree on the importance of good source documentation. Because, as the saying goes, “If it wasn’t documented, it wasn’t done.”
The International Conference on Harmonization (ICH) for Good Clinical Practice (GCP) guidance has introduced strict rules and regulations for clinical trials. Medical experts are trying to implement an international standard for research (including design, performance, and report) in order to obtain valid and credible results. At the same time, professionals need to make sure that research procedures guarantee people’s safety and confidentiality.
Note that since 1996, ICH E6 has been accepted as the Gold Standard for International Good Clinical Practice. It has become the leading international quality standard for conducting clinical trials (Wilsher, 2015). ICH E6 guidance guarantees credibility, safety, and transparency of medical research.
Researchers all over the world agree that precise, detailed, and accurate source documentation is crucial for any clinical trial. So let’s explore the meaning of ‘source documentation.’ Source documentation can be any medical record or form kept for each participant – prior, during, and after the clinical trial procedures (Bargaje, 2011).
Source documentation can consist of hospital records, notes, diaries, checklists, transcriptions, X-rays, charts, and much more. As mentioned above, records should include the time before, during, and after the study. Most of all, source documentation needs to be clear, precise, and complete.
Source documentation is the crucial link that permits investigation and validation of data at any time, by any research body. As audits are crucial, good documentation can only facilitate the process, supporting successful outcomes. Source documentation gives participants a unique chance to track the complete research journey of each participant. At the same time, good source documentation can prove that ethical rights and subjects’ well-being have been respected during research.
We should not forget that good source documentation gives researchers the exciting opportunity to reconstruct the study and add more valuable insights to the existing knowledge. Each small grain of knowledge helps researchers load the medical sack of progress.
As medical trials determine people’s physical and emotional well-being, many ethical issues are involved in research. Thus, professionals and institutions need to follow numerous strict regulations. Due to the wide range of regulations, authorities have taken a crucial step: they’ve imposed research standards.
The U.S. Food and Drug Administration (FDA) became the first body to introduce standards. In a simple form, all main aspects of good documentation have been presented. This form is known as ALCOA-C (ALCOA before). ALCOA-C includes some crucial definitions and regulations of good source documentation. In addition, it tackles all possible issues that may emerge from any electronic source documentation.
As mentioned above, ALCOA-C is the leading guidance that helps researchers and audits. ALCOA-C can be described as an insightful checklist, which is part of the official 21 Code of Federal Regulations (CFR) 58.130 (e). It has been implemented in medical practices in order to ensure confidentiality, credibility, accuracy, and validation.
Note that ALCOA-C is an abbreviation of a few crucial concepts in the field of clinical trials and good source documentation (“Clinical Research Documentation”), which researchers need to implement and follow.
Attributable: As medical records are official documents, the first ‘attributable’ principle is crucial. To be more precise, it should be clear who’s created a medical record and when. In other words, medical records, changes, and additional documentation should be attributable to researchers. Simply because research is linked to people’s safety and well-being. The same principle is valid for all changes: if a change is made in a document, that should be clear (in addition, names and dates should be stated).
To make sure these procedures are manageable, the research team needs to have a working Delegation of Duties Log (Sfera, 2016). The titles, roles, training, and education of personnel need to be listed in order to support the validation of data and audits.
Legible: This is another crucial aspect, which implies that data and medical records should be easily read. As external parties are involved, this principle should be a focus of good source documentation practices.
Let’s say that something is unreadable. Then how can researchers be sure that the data is correct? Note that binding text or errors are not acceptable as transparency is needed. Quite the opposite: If an error has been spotted, researchers should strike it with one line and rewrite it clearly, including dates and initials.
Contemporaneous: As clinical trials are prone to delays and as longitudinal observations may be needed, researchers should consider the principle of ‘contemporaneous.’ In other words, all results and changes should be made in real-time. Signatures, initials, and dates should be entered clearly (including the date of each signature).
This principle is important because it shows if data is credible or not. The documents need to show a good research story with a sensible flow and transparent narration. Otherwise, questions regarding fraud and misinterpretation may arise.
Original: Original documentation instead of photocopies are required in order to guarantee accuracy and confidentiality. This way credibility of results can be guaranteed.
Note that any item on which a data point is collected is seen as the original source document. Therefore, researchers should avoid writing down results or ideas on random items. Staff needs to be trained in using spreadsheets, medical equipment, or electronic systems. At the same time, good documentation should guarantee people’s confidentiality and safety. The research team should consider online safety and cybersecurity. The data stored in the cloud needs to be not only available but protected. In the end, patients’ rights are what research should revolve around.
Accurate: Most of all, records should be honest and accurate. Not surprisingly, medical records should be checked for errors and inconsistencies. The different pieces of equipment, research, and staff need to form a harmonic entity.
Again, we should mention the importance of entering dates and initials. These standard procedures can help investigators track errors or changes in order to guarantee the accuracy and credibility of results.
Complete: Last but not the least, researchers need to make sure that their documentation is complete. Both parties, researchers, and investigators should aim for accurate and complete source documentation. Let’s not forget that good source documentation can foster collaboration between institutions, which is crucial in the field of medical research.
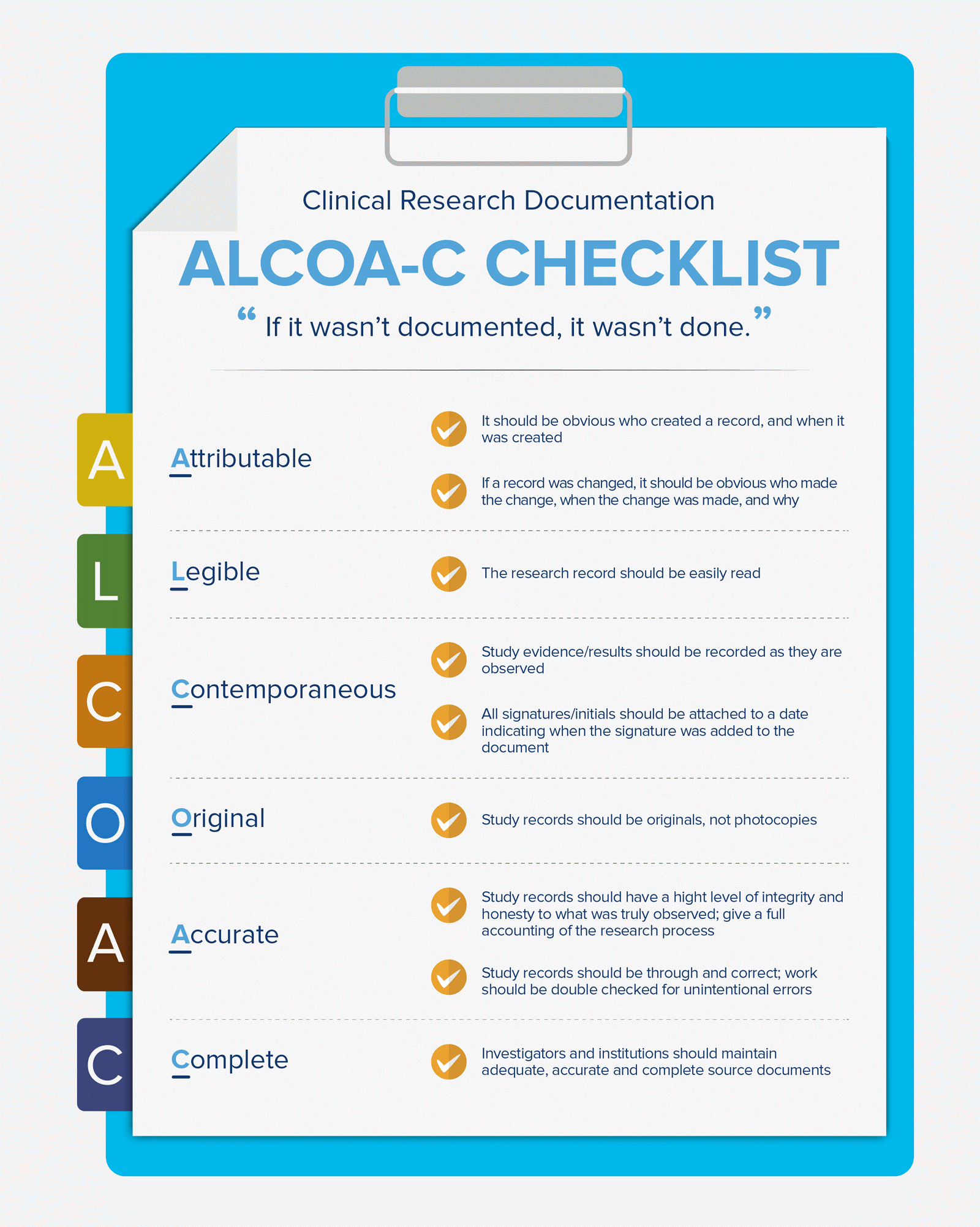
Note that originally ALCOA-C was called ALCOA. In 2010, four letters/principles were added: CCEA (Complete, Consistent, Enduring, and Available when needed). As ‘Complete’ became an integrated part of it, now, the checklist is known as ALCOA-C.
However, let’s not forget that the other three principles (Consistent, Enduring and Available when needed), should also be followed. Data should be accurate, clear, and consistent in order to present any trial as a synchronized process. Endurance is also crucial: results should provide meaningful insights for long term. Also, researchers need to make sure that data is available when needed in order to guarantee transparency and safety at the same time. Data should be easily available during audits. Of course, data should be credible and corroborated (Bargaje, 2011). In other words, data should be based on real facts and researchers should be able to back it up by evidence. As mentioned above, the roots of all evidence can be found in good documentation.
Recently, researchers have started to focus on electronic trial data systems, including cover system setup, installation, functionality testing, data collection, system security, and software upgrades.
This shift in perspectives is not surprising. It’s just a natural result of all the social and economic changes triggered by technology. The Electronic systems can provide accurate data; they can indicate clear changes in real-time; and most of all, they can guarantee transparency.
Last but not the least, we should focus on the opportunities our tech-driven society has to offer and digital health, in particular. As remote monitoring is becoming more and more popular, consistency between remote monitoring visits and source documentation is needed (Sfera, 2015). As mentioned above, electronic systems can facilitate the process of good source documentation.
To sum up, there are many challenges researchers, sponsors, and patients face. However, one thing is for sure: Good documentation practices guarantee success. Simply because even the most valuable study, creative team, and advanced technology might fail without good documentation. The documented records, procedures, forms, videos, and other research techniques/tools need to be the main aim of research and clinical trials.
So far, the ICH E6 practice and the ALCOA-C checklist have been established as the Gold Standard for International Good Clinical Practice. Thus, health experts need to implement these principles in research. Professionals need to aim for Attributable, Legible, Contemporaneous, Original, Accurate, and Complete Data, which is Consistent, Enduring, Available, Credible, and Corroborated at the same time. Most of all, results and data need to be transparent in order to facilitate audits, collaboration, and research in general.
Only by validating, questioning and replicating findings, clinical trials can lead to success. In the end, good documentation can help not only research institutions and sponsors but future medical studies. All that with the sole purpose of helping patients!
Monday – Friday
9 AM – 5 PM EST
DISCLAIMER: ConductScience and affiliate products are NOT designed for human consumption, testing, or clinical utilization. They are designed for pre-clinical utilization only. Customers purchasing apparatus for the purposes of scientific research or veterinary care affirm adherence to applicable regulatory bodies for the country in which their research or care is conducted.