
The electron transport chain (ETC) is a group of protein complexes that function in the last stage of cellular respiration.
In eukaryotes, the electron transport chain is found in the inner mitochondrial membrane where each component acts in sequence to catalyze redox reactions, transfer electrons from their donor to acceptor molecules, and simultaneously transport protons (H+) across the inner mitochondrial membrane to the intermembrane space.
This process of transferring electrons via the ETC regenerates nicotinamide adenine dinucleotide (NAD+) and flavin adenine dinucleotide (FAD+) to resupply the metabolic pool.
Moreover, the transfer of protons across the membrane also establishes the proton gradients which provide energy for oxidative phosphorylation that synthesizes adenosine triphosphate (ATP) for cellular use.
Cellular respiration is a cellular catabolic process that harvests potential energy from chemical bonds of carbohydrate molecules in the form of ATP. The complete cellular process consists of four pathways: glycolysis, pyruvate oxidation, the Krebs cycle, and oxidative phosphorylation.
When a glucose molecule enters glycolysis, it is transformed into two pyruvate molecules. With sufficient oxygen level, each pyruvate molecule is transported to the mitochondrial matrix where it is oxidized and transformed into acetyl coenzyme A (acetyl-CoA), which undergoes a series of enzymatic reactions in the Krebs cycle.
At the end of the first three cellular respiration pathways, FAD+ and NAD+ are reduced into NADH and FADH2, and ATP is generated from substrate-level phosphorylation. Though the amount of ATP generated is substantial, it is insufficient for all cellular activities. Thus, most ATP molecules are synthesized during the last stage of cellular respiration from oxidative phosphorylation.[1]
In oxidative phosphorylation, ATP is synthesized by chemiosmosis. It is connected with redox reactions, where electrons are transferred in the final stage of cellular respiration. It is mediated by a group of protein complexes in the inner mitochondrial membrane called the electron transport chain.
The electron transport chain receives electrons from NADH and FADH2 in the mitochondrial matrix, which results from the previous stages of cellular respiration. NADH and FADH2 are oxidized into NAD+ and FAD+, while the proteins in the electron transport chain are reduced.
To return to their normal state, the reduced electron transport proteins donate the electrons to the next electron carriers in the electron transport chain. Eventually, the electrons are donated to oxygen, which is the final electron acceptor in the oxidation-reduction reactions of cellular respiration. The resulting NAD+ and FAD+ are recycled to supply other pathways.[1]
The oxidation of NADH and FADH2 by the electron transport chain is coupled with chemiosmosis. During electron transfer, proteins in the electron transport chain complexes pump the protons in the mitochondrial matrix to the intermembrane space.
The accumulated protons in the intermembrane space generate proton gradients across the inner mitochondrial membrane. Since the protons are positively charged, proton gradients create an electrochemical (pH and charge) difference, which generates an electrochemical potential.
The electrochemical potential serves as a proton motive force that drives the transport of protons down the concentration gradients through ATP synthase, which situates in the inner mitochondrial membrane. The transport of protons to the matrix through ATP synthase phosphorylates ADP and synthesizes ATP in the process.
Thus, the electron transport chain establishes the proton gradients during electron transport, i.e. NADH and FADH2 oxidation. In turn, the resulting proton gradients drive the synthesis of ATP during oxidative phosphorylation.[1,2]
Also, pumping of proton by ATP synthase causes a conformational change in the protein. The protein (ATP synthase) is like a wheel and axle. So when proton is pumped through it, the coupling of ADP and inorganic phosphate occurs to yield ATP.
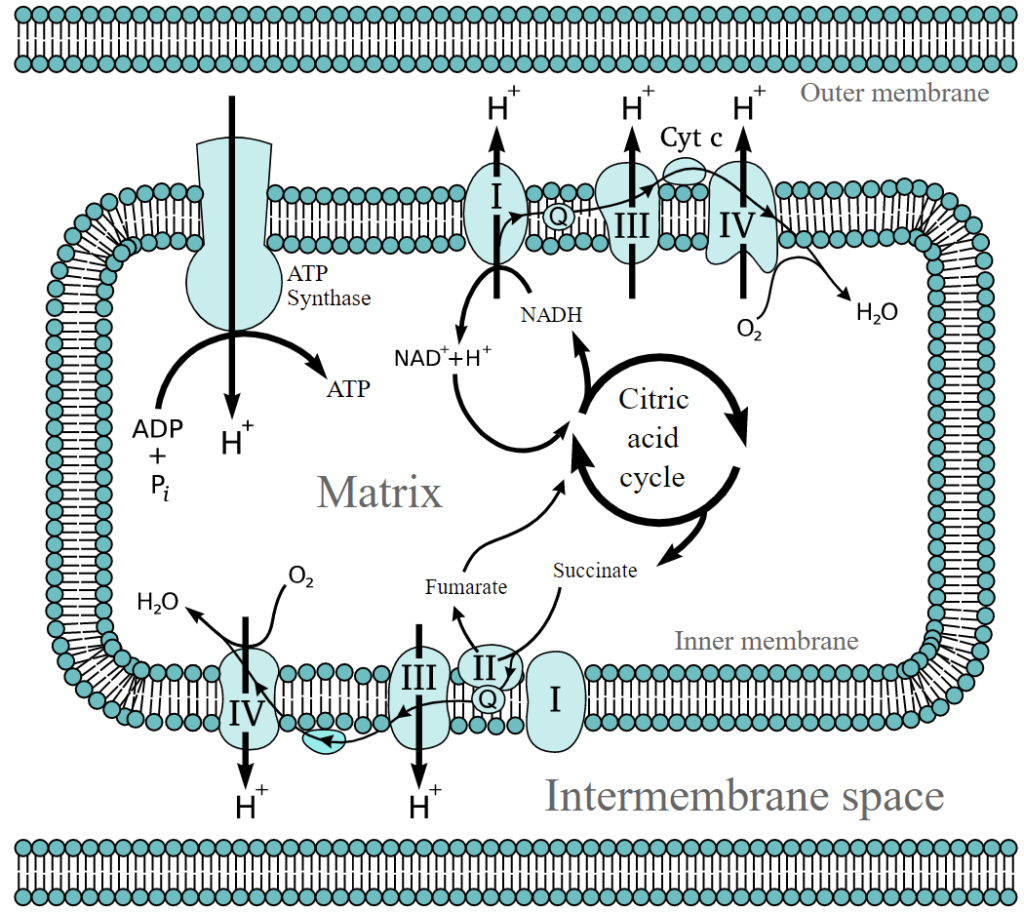
Figure 1: Electron Transport Chain and the enzyme ATP synthase.
Credit: By Fvasconcellos 22:35, 9 September 2007 (UTC) – Vector version of w:Image:Etc4.png by TimVickers, content unchanged., Public Domain, https://commons.wikimedia.org/w/index.php?curid=2716553
The electron transport chain can be divided into 4 key processes which are:
The electron transport chain consists of four multisubunit protein complexes located in the inner mitochondrial membrane. The proteins in each complex oxidize NADH and/or FADH2 and carry the electrons to the next acceptors. As the name implies, the electrons travel through a sequence of proteins in the electron transport chain until they are donated to oxygen, reducing it to water.
The protein complexes that make up the electron transport chain are:[1,2]
Also known as ubiquinone oxidoreductase, Complex I of the electron transport chain comprises of NADH dehydrogenase, flavin mononucleotide (FMN), eight iron-sulfur clusters (Fe-S), and coenzyme Q (CoQ).[3]
NADH from earlier cellular respiration stages donates two electrons to FMN. Each electron donated to FMN is transferred to the Fe-S, which is transferred to CoQ.
The fully oxidized form of CoQ is also known as ubiquinone (UQ). UQ can be reduced to ubisemiquinone (UQH) when receiving one electron and to ubiquinol (UQH2) when receiving a second electron. Subsequently, reduced UQ, UQH, or UQH2 transfer electrons to Complex III of the electron transport chain.
During electron transport in Complex I, four (4) protons are simultaneously transported from the mitochondrial matrix to the intermembrane space, setting the electrochemical gradients that drive ATP synthesis in oxidative phosphorylation.[1-3]
Moreover, the change in redox state of the protein in complex I causes a conformational change which drives pumping of protons across the inner mitochondrial membrane.
NADH + UQ + 5H –> NAD+ + UQH2 + 4H+
Most enzymes in Complex II of the electron transport chain are similar to the ones found in Complex I. However, unlike Complex I, electron transport in Complex II is not coupled with the transport of protons from the inner mitochondrial membrane to the intermembrane space.
Another major difference is Complex II’s direct connection to the Krebs cycle and, in certain cells, β-oxidation, a pathway that breaks down fatty acids into acetyl CoA.[1]
In Complex II, the enzyme succinate dehydrogenase in the inner mitochondrial membrane reduce FADH2 to FAD+. Simultaneously, succinate, an intermediate in the Krebs cycle, is oxidized to fumarate. Fumarate returns to the mitochondrial matrix, where it re-enters the Krebs cycle and undergoes a series of enzymatic reactions.
Similar to the electron flow in Complex I, the electrons from the oxidation of FADH2 are passed to Fe-S and subsequently to CoQ, reducing it to UQH and UQH2, respectively. Eventually, electrons carried by UQH2 are sent to Complex III.
Unlike Complex I, the protons in Complex II are not transported to the intermembrane space during electron transport. Thus, Complex II’s contribution to ATP synthesized from oxidative phosphorylation is considerably less than other complexes.[1,2]
Complex III of the electron transport chain consists of cytochrome b and cytochrome c1 complexes, which contain a two-iron two-sulfur cluster (2Fe-2S) called the Rieske center and a heme prosthetic group.[2-3]
Electrons that enter Complex III are carried by UQH2 from Complex I or II to Complex III. Since UQH2 carries two electrons, while the heme prosthetic group in either cytochrome can accommodate only one electron at a time, the transfer of electrons in Complex III occurs in a series of redox reactions called the Q cycle.
The Q cycle starts when the first UQH2 enters Complex III and binds to the Rieske center. There, UQH2 is oxidized into UQH, donating one electron to cytochrome c1. Subsequently, UQH is oxidized into CoQ/UQ when it donates another electron to cytochrome b.
The reduced cytochrome c1 carries the accepted electron to cytochrome c, which brings the electron to Complex IV, the last complex of the electron transport chain. In cases of reduced cytochrome b, the electron is transferred from cytochrome b to CoQ on the other side of the complex, replenishing UQH in the process.
Finally, UQH is further reduced to UQH2 when it accepts another electron from the next cytochrome b that is reduced by the second UQH2 that enters Complex III.
As a result, when two UQH2 enter Complex III, four electrons move through the Q cycle in Complex III, and one UQH2 is regenerated. Two electrons are carried to Complex IV by cytochrome c, and two electrons are used to regenerate UQH2.
Concurrently, one proton is transferred across to the intermembrane space each time an electron is donated to cytochrome c1 or from cytochrome b. Thus, four protons are pumped into the intermembrane space in one Q-cycle, adding to proton gradients across the inner mitochondrial membrane.[1-3]
In summary:
UQH2 + 2Cytc + 2H+ –> UQ + 2Cytc + 4H+
The last complex in the electron transport chain receives electrons from Complex III and transfers them to oxygen, the final electron acceptor in cellular respiration.
Complex IV consists of cytochrome a, cytochrome a3, a copper atom CuB, and a copper atom pair CuA center, which can accommodate four electrons, acting as a redox center.[2-3]
One oxygen molecule can accept four electrons. For this reason, four cytochrome c, each carrying one electron from Complex III, are required to reduce one oxygen molecule into two water molecules.
Similar to Complex I and III, two protons (for every two cytochrome c that is oxidized) from the matrix are transported across the inner mitochondrial membrane to the intermembrane space during electron transport.[3]
2Cytc + ½O2 + 4H+ –> 2Cytc + H2O + 2H+
The accumulation of protons builds on the existing proton gradients that occur during electron transport in Complex I and III. The proton gradients add to the existing electrochemical potential, which provide the proton motive force that drives ATP synthesis in oxidative phosphorylation.
It is estimated that the complete oxidation of one NADH molecule would result in three molecules of synthesized ATP, while the complete oxidation of one FADH2 molecule will yield two molecules of synthesized ATP.[1-3]
The electron transport chain is a group of protein complexes that facilitate the transfer of electrons in the final stage of cellular respiration. They catalyze redox reactions that resupply the cellular NAD+ and FAD+ pool while setting up the electrochemical potential that is essential for the synthesis of ATP by oxidative phosphorylation.
Malfunctioning electron transport chains can result in depleted energy levels and the formation of radicals such as reactive oxygen species. While this can affect the health and well-being of the cells, the understanding of such disturbances is useful and applicable in agricultural practices and drug designs.

Monday – Friday
9 AM – 5 PM EST
DISCLAIMER: ConductScience and affiliate products are NOT designed for human consumption, testing, or clinical utilization. They are designed for pre-clinical utilization only. Customers purchasing apparatus for the purposes of scientific research or veterinary care affirm adherence to applicable regulatory bodies for the country in which their research or care is conducted.