Amino Acids: Building Blocks of Proteins

Reference to this article: ConductScience, Amino Acids: Building Blocks of Proteins (2022). doi.org/10.55157/CS20220612
Introduction and History
Amino acids are organic compounds consisting of carbon, hydrogen, and nitrogen. They are monomers or building blocks of proteins. Proteins are one of the major biomolecules required for the proper functioning of living organisms. Moreover, they are the first biomolecules that were first recognized for their biological roles in organisms than the other biomolecules including carbohydrates, nucleic acids, and lipids.[1]
Moreover, proteins have well-defined physicochemical properties. The linearly arranged amino acid sequence, or one-dimensional protein, guides the formation of the 3-D structure of the protein that determines its interaction with other molecules.[1]
It’s been a century since the observation that when proteins are exposed to hydrolytic actions of boiling acids, they split up into small molecular substances.[2] The amino acid, Glycine, was first discovered and isolated by scientists from gelatin. Soon after this discovery, other amino acids were unmasked including cystine, asparagine, leucine, and threonine — the last of the 20 common amino acids to be found, in 1935.[2]
Several studies led to the discovery of about 500 amino acids by 1983. But, only 20 were found to be present in the genetic code and considered to be the essential building blocks of proteins.[1]
This article brings you an overview of amino acids (monomeric units of proteins), their structures, functions, and applications in the real world.
Classifications of Amino Acids
1. Classification based on the nutritional requirement
Out of 20 amino acids, 9 can be synthesized by the human body, and the others are gotten through the food we eat. These criteria categorize these amino acids into two groups: Essential amino acids and non-essential amino acids.
1.1. Essential amino acids
The amino acids not synthesized by the body and required from dietary sources are called essential amino acids. It includes histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine.[4]
They can be obtained from various food sources such as quinoa, egg, meat, chicken, and vegetables.[4] These essential amino acids help the body repair muscle tissues and form precursor molecules for neurotransmitters.
1.2. Non-essential amino acids
The amino acids synthesized by the body are called non-essential amino acids. They are involved in proper brain function, the production of red blood cells (RBCs) and white blood cells (WBCs), and the removal of toxins from the body. It includes arginine, aspartic acid, glutamic acid, asparagine, glutamine, glutamic acid, proline, glycine, serine, and tyrosine.[5]
Even though they are produced by the body from scratch, the dietary sources containing these amino acids can enhance their availability and functions in the body.[4]
2. Classification based on the polarity of the side chain
The amino acid structure is composed of an amino group and a carboxyl group along with a side chain R. All amino acids have different side chains, as a result, they show different physicochemical properties and functions.[1] The key elements of the amino acid structures are carbon (C), hydrogen (H), oxygen (O), and nitrogen (N).
All the amino acids, except proline, are known as alpha-amino acids. Proline contains a secondary amine where an alpha carbon is attached to two other carbons. Thus, it is referred to as the alpha-imino group.
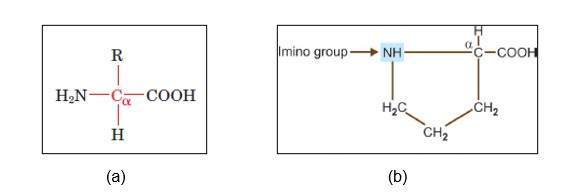
Figure: (a) General structure of alpha-amino acids, and (b) structure of proline showing secondary carbon and imino group.[1]
The amino acids are categorized into three groups based on their side-chain structures:
2.1. Amino acids with nonpolar side chains
Nine amino acids have a non-polar side chain in their structures. These include glycine, alanine, valine, leucine, isoleucine, methionine, proline, phenylalanine, and tryptophan.
Glycine has the smallest side chain with an H atom; alanine, valine, and leucine contain an aliphatic hydrophobic side chain; methionine contains a thiol ether side chain; proline has cyclic secondary amino acids; phenylalanine has a phenyl moiety; while tryptophan has a bulky indole aromatic side chain.[3] The aliphatic or aromatic groups attached to these amino acids make them hydrophobic (repel water).
While building proteins, the globular proteins bury these hydrophobic amino acids in the interior of their 3-D shape to prevent them from any water contact.[6]
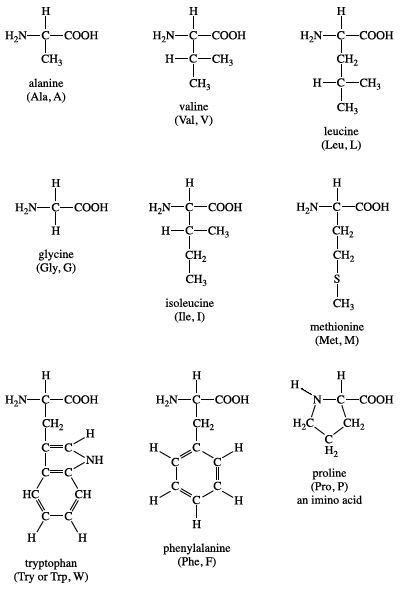
Figure: Structure of non-polar amino acids.[6]
2.2. Amino acids with polar side chains
Six amino acids fall in the category of uncharged polar side-chain amino acids. These include serine, threonine, asparagine, glutamine, tyrosine, and cysteine.
The amino acids, serine, and threonine contain aliphatic hydroxyl groups of different sizes; glutamine and asparagine have amide groups of different sizes; tyrosine has phenolic group, an aromatic group like tryptophan and phenylalanine; and cysteine has a unique thiol group attached to it.[3]
These amino acids have available electron pairs that are involved in hydrogen bonding with water and other molecules.[6]
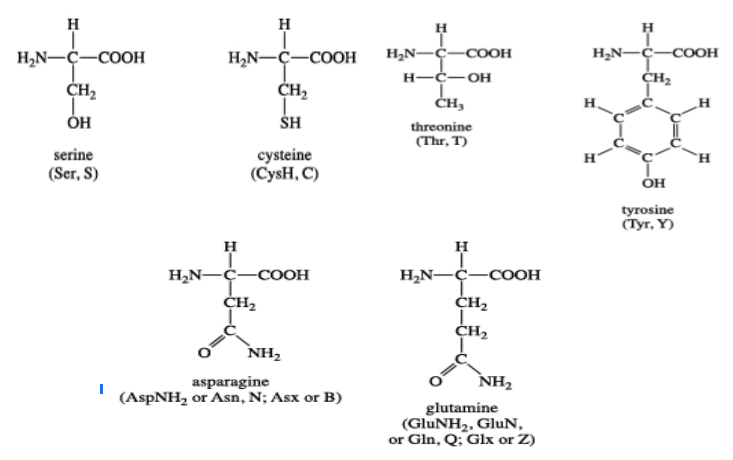
Figure: The structure of polar uncharged amino acids.[4]
2.3. Amino acids with charged polar side chains
The side chain of these amino acids can be positively charged or negatively charged at physiological pH. It includes lysine, glutamic acid, aspartic acid, histidine, and arginine.[3]
Lysine contains a butylammonium side chain; arginine has a guanidino group; histidine bears imidazolium moiety; glutamic acid and aspartic acid carries a carboxylic acid group on their side chain.[3]
In an aqueous solution, at physiological pH, aspartic acid and glutamic acid carry an overall charge of (-1) and are called glutamate and aspartate in their ionic forms. These are also known as acidic amino acids.[6] Whereas, lysine, histidine, arginine possess a (+1) charge at their physiological pH and are called basic amino acids.[4]
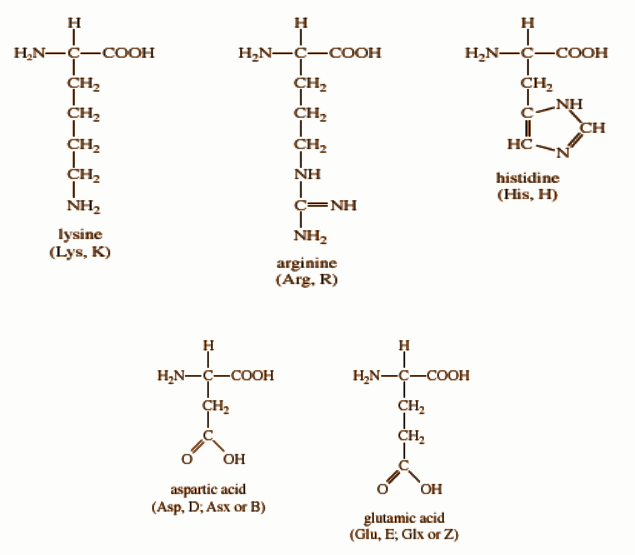
Figure: Structures of polar and charged amino acids.[4]
Physicochemical Properties of Amino Acids
The amino acids possess different physicochemical properties. These include optical activity, polarity, acidity and basicity, aromaticity, confirmation flexibility, and chemical reactivity. Some of the properties are discussed here in brief.
1. Stereochemistry
All the amino acids exist in L-configuration and are superimposable mirror images of each other. This configuration was given by Emil Fischer.[3] L- configuration represents all amino acids that are majorly involved in the translation process for protein synthesis. In some rare cases, D-amino acids are also found in some proteins.
According to the Fischer convention, the configuration of a group around a chiral center can be related to that of glyceraldehyde structure. For alpha-amino acids, the arrangement of the amino, carboxyl, R, and H groups about the C atom is related to that of the hydroxyl, aldehyde, CH2OH, and H groups, respectively, of glyceraldehyde.[3]
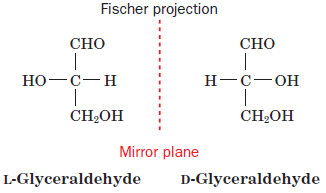
Figure: Fischer convention configuration of glyceraldehyde to which all groups can be related.[3]
2. Peptide bond formation
The number and type of amino acids linked together to form proteins ultimately determine the shape, size, and function of the proteins.[7] The amino acids are covalently attached through a peptide bond, which is a result of a condensation reaction.
The carboxyl group of one amino acid combines with the amino group of another amino acid releasing a water molecule, this reaction is called a dehydration or condensation reaction.[7]
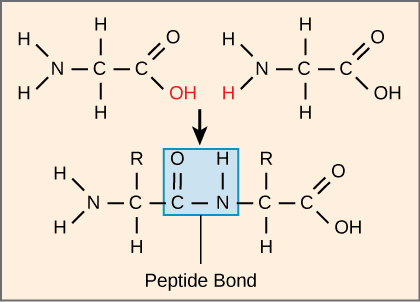
Figure: Peptide bond formation between two amino acids with the release of a water molecule.[7]
Less than 50 amino acids linked together with the peptide bond are called oligopeptides and more than 50 amino acids are combined to form polypeptides. So, the proteins are the result of polypeptide formation, whereas individual amino acids are known as amino acid residues.[6]
It’s also essential to note that an individual amino acid possesses acid-base properties, which are lost in proteins because of the involvement of the carboxyl group in the peptide bond formation. So, the acidity-basicity of proteins depends on the overall ionization characteristics of individual R groups of the amino acid components.
3. Optical properties
Amino acids, except glycine, isolated after mild hydrolysis of protein can rotate plane-polarized light. This property is known as optical activity. The optically active molecules possess chiral carbons, having four different groups attached to a carbon, called a chiral center.[3]
The asymmetry due to chiral carbons results in non-superimposable mirror images of the molecule, called enantiomers. Glycine has two hydrogens attached to its alpha-carbon and is therefore not optically active.
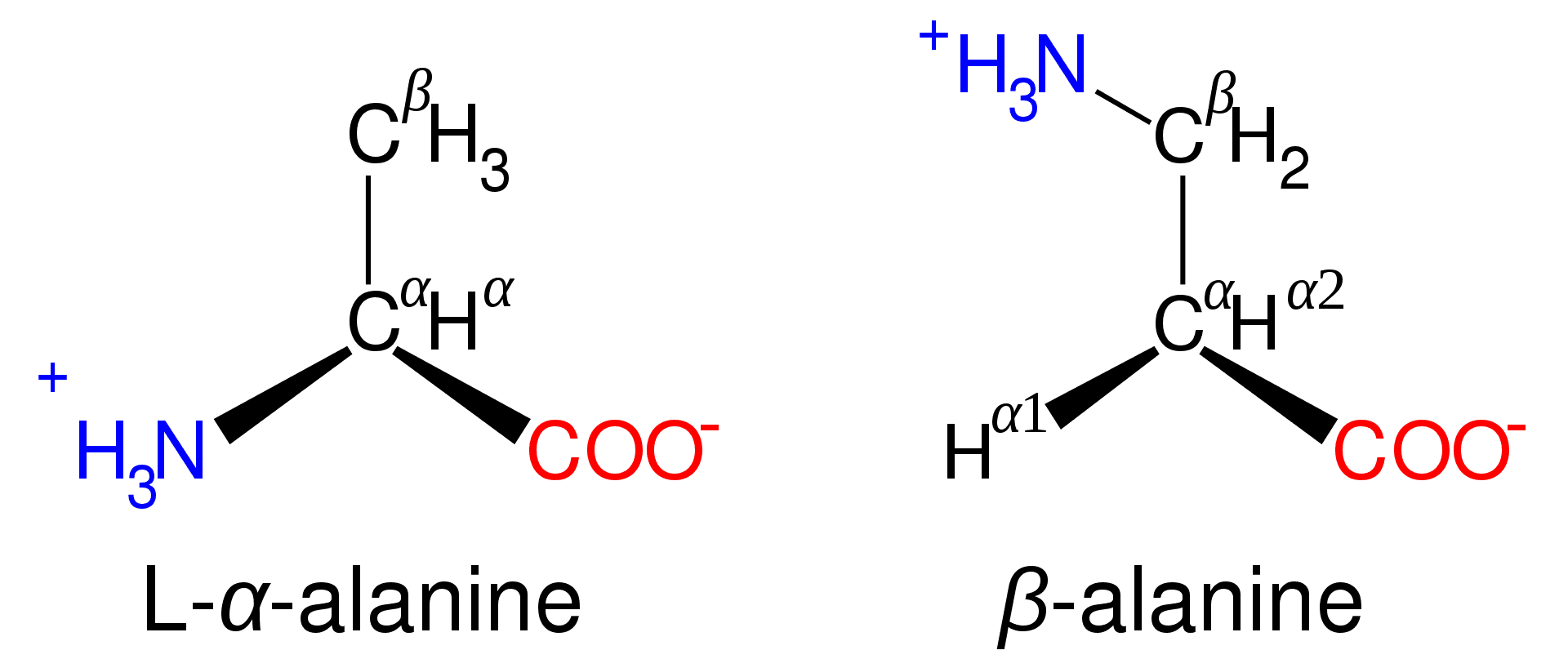
Figure: Structure of alanine isomers.[1]
The concept of enantiomers should not be confused while performing ordinary chemical synthesis. The chemical or physical processes have no stereochemical bias, thus, during the chemical synthesis of chiral molecules, racemic mixtures are produced.[3] Racemic mixtures contain an equal amount of right-hand and left-hand enantiomeric forms of a molecule.
4. Acid-base properties
Amino acids are amphoteric molecules, as they can either act as an acid or a base. The acidity and basicity of amino acids are due to charges either on their carboxyl or amino groups.[6]
The pKa value of the basic amino group is between 9 and 10 while that of an acidic carboxyl group is close to 2. The pKa value indicates the strength of amino acids. It is defined as the negative base (-10) log of the acid dissociation constant (Ka) of a solution.
pKa = -log10Ka
The pKa is also defined as the pH value at which the concentration of protonated group is equal to that of the unprotonated group.[6] So, at physiological pH, the amino acids have a balanced charge, equal number of negative and positive charges, on their structure and are called dipolar ion or zwitterion. The pH at which this phenomenon occurs is called the isoelectric point, donated by pI.
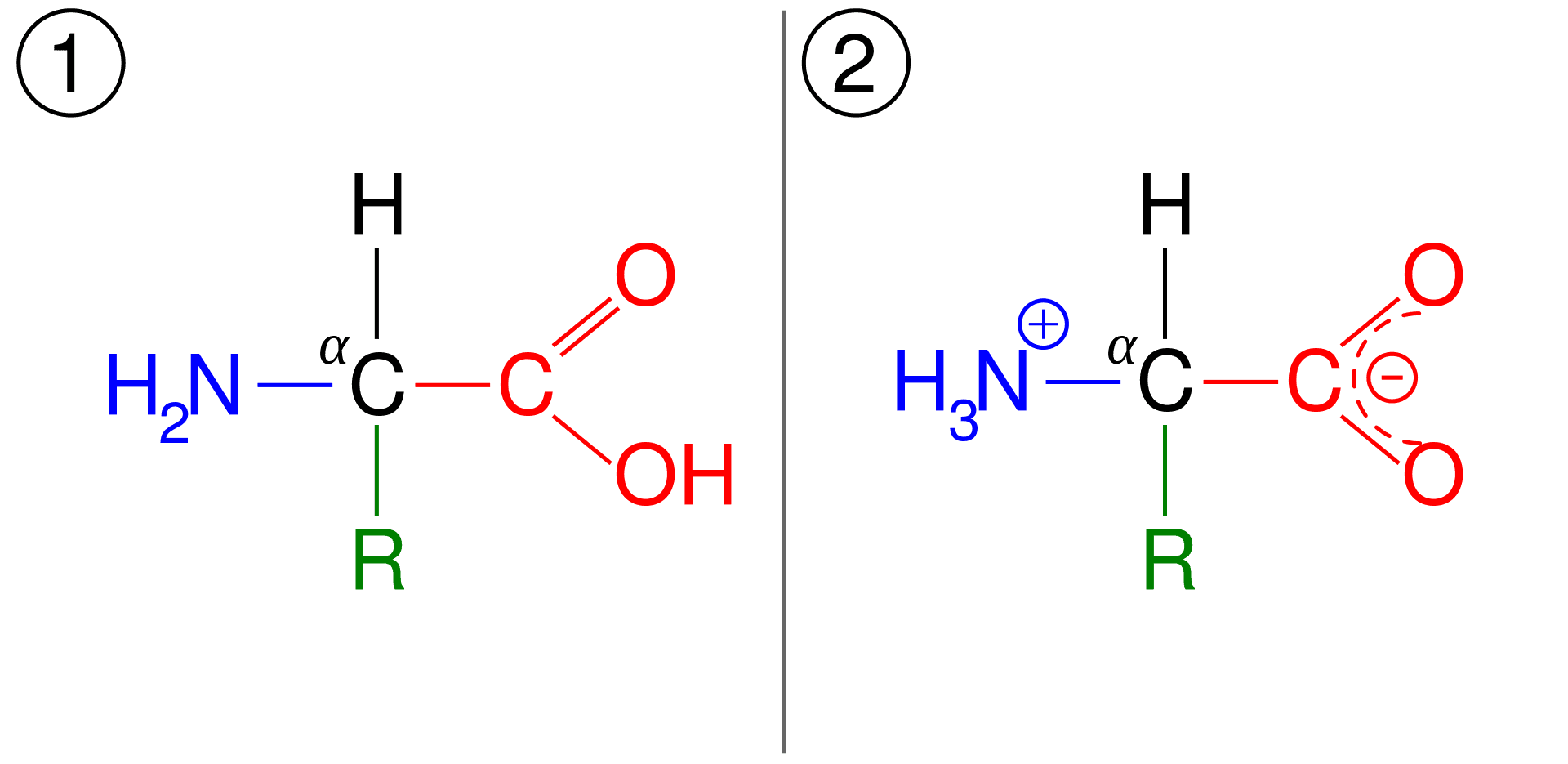
Figure: (1) General structure of amino acid, and (2) its zwitterionic form.[1]
Note: At the isoelectric point, amino acids have a net-zero charge but amino acids are never considered to have an absolute zero charge.[6] Moreover, in an aqueous solution, amino acids never assume a neutral form.
5. Chemical reactions
The amino acids undergo several reactions by involving their functional groups. Some chemical reaction processes of amino acids are given below:
- Deamination is the transfer of the amino group of one amino acid to a compound that forms another amino acid. For example, the transfer of an amino group to alpha-ketoglutarate results in glutamate formation.[1]
- The Condensation reaction is the combination of two components into a single compound while releasing a small molecule. Two or more amino acids undergo a condensation reaction to form polymers of amino acids by forming a peptide bond and releasing one or more water molecules at the same time.[4]
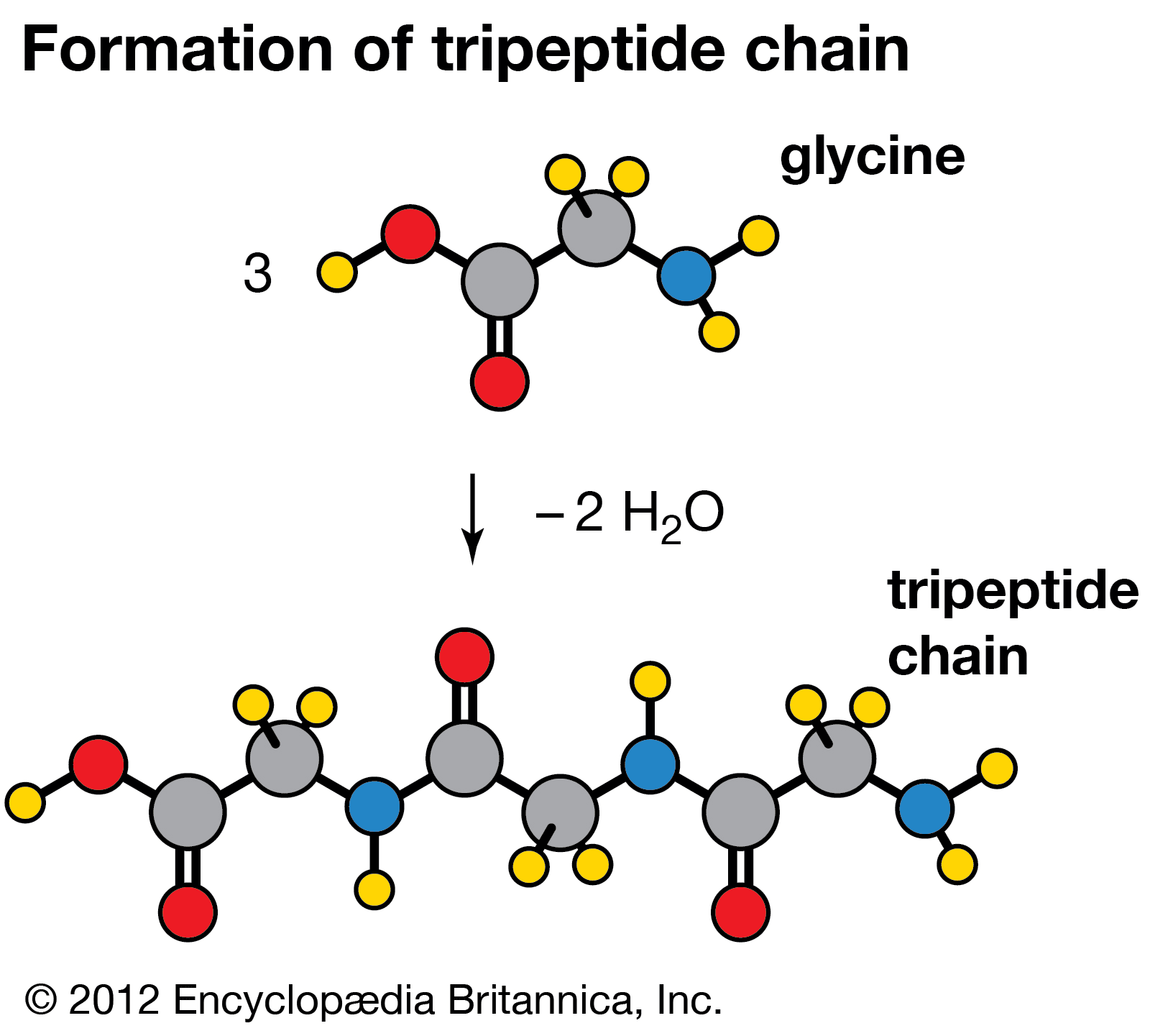
- Figure: Condensation reaction of three molecules of glycine into a tripeptide chain by releasing two molecules of water.[8]
- Cysteine oxidation is the oxidation of two molecules of cysteine that result in cystine production by forming disulfide bonds. The reaction occurs due to the high reactivity of the thiol group of cysteines that are involved in this reversible oxidation reaction.[4] The reaction is mainly essential for proteins. The disulfide bridges formed in the reaction provide stability to proteins.
Functions of Amino Acids
All amino acids have some central role to play in the body’s metabolism and functioning. So, given below is a list of essential functions performed by some amino acids:
- The thiol side chain of the cysteine is involved in the formation of a unique disulfide bond by cross-linking with the thiol group of another cysteine. This bonding has a crucial role in protein formation and stability.[3]
- Threonine plays a major role in signal transduction and boosting the immune system.[8]
- Phenylalanine is a precursor molecule in the synthesis of tyrosine, dopamine, norepinephrine, epinephrine, and melanin. The genetic disorder, phenylketonuria, is caused due to an inability to synthesize and metabolize phenylalanine.[8] This is because of a lack of phenylalanine hydroxylase.
- Tryptophan helps in anchoring cell membranes along with tyrosine. They are also precursor molecules for the production of neurotransmitters (serotonin), hormones (melatonin), and the vitamin, niacin.[8]
- Alanine plays a crucial role in the glucose-alanine cycle between tissue and liver. This cycle removes and transfers pyruvate and glutamate from muscle to liver. An imbalance in the glucose-alanine cycle increases the level of ALT (Alanine transferases) which leads to type II diabetes.[8]
- Valine is essential for the self-renewal of hematopoietic stem cells that give rise to other blood cells.[8]
- Leucine is a ketogenic molecule that gives an end product of acetyl CoA and acetoacetate.[8] These molecules are precursors of ketone bodies and myelin that are especially required during early childhood for brain development.
- Isoleucine has multiple functions including assisting wound healing, detoxification of nitrogenous wastes, stimulating immune function, and promoting secretion of several hormones.[8]
- Methionine is a precursor for compounds like cysteine and taurine, versatile compounds such as S-adenosyl methionine, and the antioxidant, glutathione. Methionine is regenerated in the body by using homocysteines, and improper conversion leads to the accumulation of homocysteine that causes atherosclerosis.[8]
- Histidine is a precursor for histamine that plays a major role in inflammation. It is converted into ammonia and urocanic acid by the action of the enzyme histidine, ammonia-lyase.[8] The deficiency of this enzyme causes a rare metabolic disorder, histidinemia.
- Lysine is involved in protein stability, epigenetic regulation by histone modification, structural proteins of connective tissues, calcium homeostasis, and fatty acid metabolism. Any interruption in lysine catabolism results in lysine accumulation that causes hyperlysinaemia.[8]
Applications of Amino Acids
Amino acids have a variety of application in industries, some of them are listed below:
- They are used as additives in animal feed. Examples include lysine, methionine, threonine, and tryptophan.[1] The amino acids are chelated with the metal cation to improve the absorption of mineral supplements for the better health of animals.
- Aspartame (aspartyl phenylalanine 1-methyl ester) is used as an artificial sweetener worldwide.[1]
- Glutamic acid has a major application as a flavor enhancer.[1]
- Amino acids are also used as supplements to improve mineral absorption in the human body, especially for those dealing with mineral deficiencies.[1]
- Amino acids are used to manufacture drugs and cosmetic products.
- Amino acids are used to develop fertilizers to enhance mineral absorption in plants and prevent deficiencies. Thus, facilitating healthy production of plants without affecting overall productivity.[1]
Amino acids are used to manufacture biodegradable polymers that are used for developing eco-friendly packaging, drug delivery carriers, and prosthetic implants.[1]
Conclusion
Amino acids are building blocks of proteins that are involved in every metabolic function of organisms. The side chain of the amino acids determines their physical and chemical properties. The chemical nature of these amino acids is determined by their functional groups. These functional groups are involved in carrying out several chemical processes in the organisms.
Today, several tools are present to isolate and study amino acids in labs. The advanced technology has facilitated the sequencing of every protein to study from their basic unit, that is, the amino acid. Scientists are meticulously developing several other tools to tackle the challenges faced during the study of these molecules. The area of amino acid and protein research creates an intriguing possibility to discover and uncover several other mysterious properties of these amino acids and their functions in organisms.
References:
- Amino acids. Retrieved from https://en.wikipedia.org/wiki/Amino_acid
- Vickery, H. B., & Schmidt, C. L. A. (1931). The History of the Discovery of the Amino Acids. Chemical Reviews, 9(2), 169–318. doi:10.1021/cr60033a001
- Voet Donaly and Voet G. Judith (2010). Biochemistry. Hoboken, NJ: John Wiley & Sons.
- Sarah Knapp (2020). Amino acids. Retrieved from https://biologydictionary.net/amino-acids/
- Difference between essential and non-essential amino acids. Retrieved from https://www.vedantu.com/biology/difference-between-essential-and-nonessential-amino-acids#
- Amino acid: Chemical compound. Retrieved from https://www.britannica.com/science/amino-acid
- Amino acids. Retrieved from https://courses.lumenlearning.com/introchem/chapter/amino-acids/
- Kiran Meena (2019). Structure of amino acids. Retrieved from https://aiimsrishikesh.edu.in/aiims/document/Biochemistry/Structures%20of%20Amino%20Acids.pdf
