The biological significance of proteins was recognized more than two centuries ago.[1] They were considered the primary material required for living organisms. But from the beginning of the 20th century, the significance of protein-like molecules, peptides, became apparent in several life processes.[1]
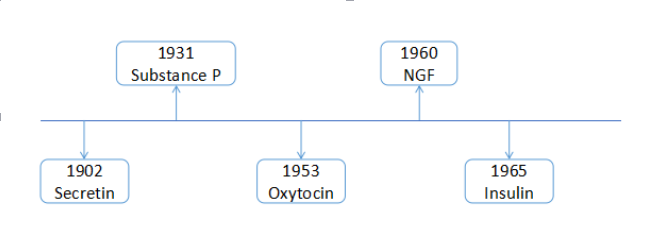
Figure: The illustration of name and year of peptides discovered in the 20th century.
Source: Creative Peptides[2]
Like proteins, peptides also consist of chains of amino acids and are held together by peptide bonds. Peptides have the same properties as protein molecules.[2] However, unlike proteins, their classes of molecules are small, simpler, and of lower molecular weight. They consist of 2-50 amino acids, unlike proteins which consist of more than 50 amino acids.[2]
Emil Fischer is considered the father of the chemistry of peptides. He coined the term “peptide,” which originated from the word “pepsis,” meaning digestion products of proteins.[1]
This article poses a classification of peptides, their classes, actions, and essential peptide functions in living organisms.
Peptides are formed by linking two or more amino acids through an amide linkage, called a peptide bond.[3] The formation of peptide bonds occurs by the removal of a hydroxyl group (-OH) from one alpha-amino group and hydrogen (-H) atom from another alpha-amino group, forming a water molecule (H2O). This reaction is called a dehydration reaction.[3]
Peptides are sub-categorized into two groups based on the number of amino acids present in their structures: oligopeptides and polypeptides.[3]
When two or more (but less than 20) amino acids are linked together with the loss of a water molecule, they are called oligopeptides. It also includes dipeptides, tripeptides, tetrapeptides, and pentapeptides. Some examples of naturally occurring oligopeptides are microviridin, cyanopeptolin, microcystins, etc.[4]
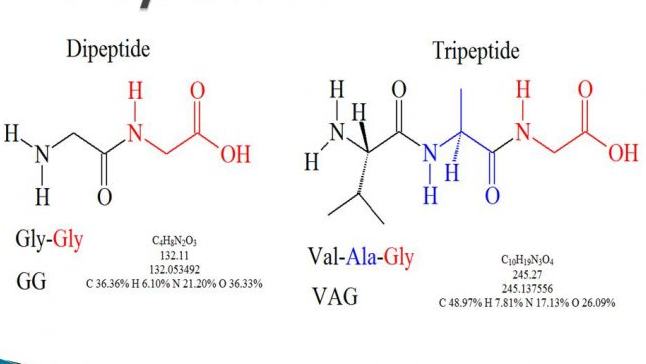
Figure: An illustrative structure of dipeptide and tripeptide.
Source: Online Biology notes.[3]
The oligopeptides are synthesized through non-ribosomal pathways, except for cyclamates and microviridin synthesized through ribosomal pathways.[4] There are several ways to identify peptides, and these ways include gel chromatography, HPLC, HPLC-mass spectroscopy, and ion-exchange chromatography.[4]
When 20 or more amino acids are linked together through covalent peptide bonds, they are called polypeptides.[4] One or more polypeptides are involved in the formation of proteins. They have two terminals present in their structure: N-terminal containing an amino group and C-terminal containing a carboxyl group. Some examples of polypeptides include insulin and growth hormones.[4]
Polypeptides are arranged in different structural forms to create different functional proteins. So, according to the number and arrangement of polypeptides, the structure of proteins are categorized into four groups:[4]
Peptides, based on their functional properties, are categorized into many small groups. Here’s a list of the most commonly studied classes of peptides in organisms, along with their functions and some examples.[5]
Antimicrobial peptides, also known as host defense peptides, are a class of peptides that play a role in the innate immune response of all organisms.[6] They are classified into two groups: ribosomally synthesized peptides and non-ribosomally synthesized peptides.[6]
As the name suggests, bacterial peptides are fragments of proteins produced by bacteria. They include flagellar peptides, lipoproteins, enterotoxins, and several enzymes.[7]
The peptides secreted from both gram-positive and gram-negative bacteria are both cationic and neutral.[6] These types of peptides are included within bacteriocin that kills specific competitor bacteria, protecting the host bacterium.[6]
Examples include Escherichia coli 7-amino-acid peptide microcin C7 (inhibits protein synthesis), Lactococcus peptide mersacidin (inhibits peptidoglycan biosynthesis), nisin, and epidermin (permeabilizes target cell membrane).[6]
These are small proteins synthesized by neurons to act on receptors and modulate synaptic transmission.[8] The neuropeptides are synthesized by large, inactive precursor proteins, called pre-propeptides.
These proteins are cleaved into several active peptides and produce multiple copies of different neuropeptides.[8] Most neuropeptides act on G protein-coupled receptors (GPCRs) and fall into two families: the rhodopsin-like and the secretin families.
Examples include acetylcholine, epinephrine, norepinephrine, dopamine, serotonin, and Gamma-aminobutyric acid (GABA).[8]
Anticancer peptides (ACPs) are small peptides with a short amino acid sequence that are selective and toxic to cancer cells.[9] The predominant amino acids in anticancer peptides include glycine, lysine, and leucine.[9]
The anticancer peptides are a highly preferred choice among all the other available anticancer therapeutics due to their high selectivity, high penetration, and easy modifications.[9]
The peptides destroy cancer cells via apoptosis and necrosis by lysing or forming pores in the membranes of cancerous cells. These types of peptides, depending on their structure, mode of action, selectivity, and efficacy to specific cancer cells, are divided into three categories:[9]
They are short amino acid-chained peptide hormones synthesized and secreted by specialized cells in the endocrine.[11] They are stored in membrane-bound secretory vesicles, which enable their rapid secretion whenever required.
They are water-soluble, and this makes it difficult for them to cross the hydrophobic cell membranes.[11] Thus, they need specific receptors on the cell surface to exert their actions.
Some examples of endocrine peptides include:
The endogenous and exogenous opioids exert several physiological and pharmacological effects through receptors of four different subtypes.[12] It include μ (μ1, μ2), 𝜹 (𝜹1, 𝜹2), 𝜿 (𝜿1, 𝜿2), and ε (ε1, ε2).[12]
They regulate other endocrine systems like the hypothalamic-pituitary-adrenocortical axis and the phenomenon of stress-induced analgesia.[12]
Endogenous opiate peptides are better studied than exogenous peptides. They consist of three families of peptides based on their origin:[12]
Some peptides like cholecystokinin (CCK), neuropeptide FF (NPFF), and melanocyte inhibiting factor (MIF)-related peptides possess anti-opioid properties.[12] But these peptide families also include some peptides harboring opioid-like properties, and that’s why they are also known as “opioid modulating” peptides.[12]
Plant peptides are peptides that originated in plants and they possess significant health benefits in humans. They lower blood pressure and cholesterol levels and inhibit enzymes within the renin-angiotensin-aldosterone system (RAAS).
Other benefits include their anti-inflammatory activity, anticancer and immunomodulatory activities, prevention of and protection against oxidative damage through free radical scavenging activities, and antimicrobial activity.[13]
Plant peptides are categorized into three groups based on their functional response:[13]
Toxins developed in animals are strategies to protect themselves from different predators and/or capture their prey.[14]
Several of these venoms studied are found in animals with envenomation apparatus like cone snails, spiders, scorpions, snakes, the Gila monster lizard, and sea anemone.[14]
Venom peptides act as natural ligands of ions channels and different receptors they bind to initiate physiological responses.[14] Based on these characteristics, they are categorized into eight groups:[14]
The application of these peptides in healthcare sectors demands many issues associated with their safety, pharmacokinetics, and delivery to be addressed.[13]
Peptides are a class of biological molecules that have essential roles in fundamental physiological processes and are required for many biochemical processes. They are small molecules made by sequential arrangements of 2-50 amino acids. The difference between peptide and protein is that proteins are large chains of amino acid sequence (50 or more) with different specialized structures.
The benefits of peptides have established a special place in the pharmaceutical landscape.[15] And the innovations in peptide therapeutics are believed to rise in the future by expanding into new indications and molecular targets, exploiting novel chemistry strategies to broaden molecular diversity, and engineering enhanced pharmaceutical properties.[15]
Further, the study of peptide benefits and chemistry opens a door for scientists to unravel its hidden potentials. It can be identifying novel targets and receptors of peptides, the discovery of more peptides in organisms and their functional properties, or the discovery of sustainable peptide-based drugs.[15]
Get the entire package for up to 50% discount with our Replication program.
DISCLAIMER: ConductScience and affiliate products are NOT designed for human consumption, testing, or clinical utilization. They are designed for pre-clinical utilization only. Customers purchasing apparatus for the purposes of scientific research or veterinary care affirm adherence to applicable regulatory bodies for the country in which their research or care is conducted.