

As an Amazon Associate, ConductScience Inc. earns revenue from qualifying purchases
Autoradiography is an imaging technique that uses radioactive sources contained within the exposed sample. In vitro autoradiography methods involve the isolation of cellular components such as DNA, RNA, proteins or lipids, followed by labeling with suitable radioisotopes. In in vivo autoradiography, radioisotopes are coupled with radioactive tracers and administered orally or via injection, and the distribution of radiation is evaluated in thin tissue or whole-body cryosections. In vivo autoradiography using laboratory animals is widely used in metabolic studies, disease monitoring and new drug development experiments. Common radioisotopes in autoradiography are sulfur-35, hydrogen-3, carbon-14, 125-iodine or phosphorus-32 (35S, 3H, 14C, 125I and 32P, respectively) which are used to determine the distribution of the radiolabeled molecules in tissues, cells or cellular organelles (Figure 1), but also in the study of protein modifications and DNA / RNA sequencing (Figure 2) (Solon, 2015).
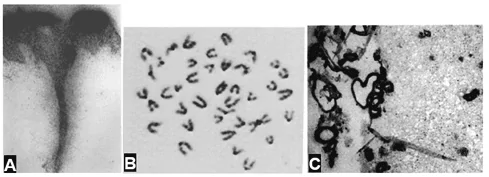
Figure 1 A – biopsy of human skin after topical application of an aqueous solution of 32P. Cut perpendicular to the epidermal surface (Witten, Brauer, Loevinger, & Holmstrom, 1956); B – 3H-thymidine labeled human chromosomes (Mukherjee, 1976); C – 125I-labeled biotin bound to amyloid plaques in frozen sections of Alzheimer disease mouse brain (Kurihara & Pardridge, 2000).

Figure 2: General principle of autoradiography. A silver halide (AgX) is ionized by the radiation emitted from radioisotopes, forming Ag+ ions. Ag+ is then reduced and converted to metallic Ag by a developer reagent (usually containing AgNO3), which precipitates within the gelatin emulsion of the X-ray film. The reduction and precipitation are stopped by emerging the film in a fixative solution forming the final image.
The principle of autoradiographic imaging is the precipitation of silver (Ag) atoms, resulting from the ionization of a silver halide (AgX – silver bromide, chloride, iodide, or fluoride – AgBr, AgCl, AgI, or AgF, respectively) by radiolabeled samples. AgX is light sensitive compound commonly used in photography. They are generally suspended in a gelatin photographic emulsion. Each AgX molecule is individually encapsulated in the gelatin, and functions as an independent detector of radioactive decay from the radiolabeled sample. Once radioactive particles hit the gelatin emulsion, AgX is reduced resulting in the production of insoluble silver crystals.
Gelatin photographic emulsions are used to coat photographic and X-ray films, which are made of a flexible base (usually cellulose acetate). When a radiolabeled sample is in contact within a coated X-ray film (exposure), it generates a latent (hidden) image corresponding to the radioactivity distribution within the sample. To make the image visible, the exposed photographic / X-ray film must be submerged in a developing reagent, a chemical mixture that converts the silver crystals into metallic silver, darkening the gelatin emulsion. Silver nitrate (AgNO3) is highly efficient in the reduction of AgX molecules and is usually a component of developer solutions. The reaction is then stopped by a fixative reagent, which removes the excess AgX from the photographic / X-ray film. Highly radioactive areas (e.g. areas with a higher concentration of a radiolabeled drug, or with higher metabolic activity) reduce more AgX molecules, resulting in higher optical density in the film (darker areas) (Figures 1 and 2) (Cagampang, Piggins, Sheward, Harmar, & Coen, 1998; Klein et al., 2016; Srinivasan, Krebs, & RajBhandary, 2006). Thus, autoradiography must be avoided in samples that are homogeneously labeled. Although it can be quantitative, autoradiography can be a slow process, depending on the half-life of the radioisotopes used.
In a clinical context, advanced autoradiographic methods like positron-emission tomography (PET) and single-photon emission computerized tomography (SPECT) are used in the diagnosis, staging, and monitoring of disease. PET and SPECT rely on the same principle as classic autoradiography but use different radiotracers (suitable for human use) and radioactive decay is detected by gamma cameras (PET / SPECT detectors). Computer algorithms are then used to produce high-resolution 3D reconstructions (tomograms) of the whole body or parts of an organism.
Classic autoradiography techniques are performed according to the following general sequential steps:
When analyzing histological sections, a counterstaining step can be added to the protocol, to identify specific tissue structures or cellular components. For example, hematoxylin and eosin staining can be used to define the nucleus and cytoplasm of cells in tissue sections, after autoradiographic detection.
In both in vivo and in vitro autoradiography, the exposure of the radiolabeled sample to AgX-containing photographic emulsion must always be performed in the dark (e.g., in a dark room, or in closed boxes). Because the AgX is sensitive to light and radiation, this ensures that the only AgX molecules reduced are the ones where radioactivity is emitted from the sample. The resolution of the autoradiographic image is proportional to the ionization capacity of the radioisotopes used. Low energy radioisotopes such as 3H reduce only neighboring silver halide molecules, producing a very sharp image, while high energy isotopes such as 125I can reduce a lot more halide molecules, producing darker images with lower specificity.

Figure 3: In vitro autoradiography for DNA analysis (www.istudy.pk)
Whole-body autoradiography (WBA) and Microautoradiography (MARG) – In vivo autoradiography techniques used to determine the tissue distribution of radiolabeled compounds in laboratory animals (Figure 4) (Solon, 2015). A radiotracer (usually labeled with 14C and/or 3H) is administered and incubated for specific time points. WBA and MARG are especially suitable for the study of receptor biology, as the radioisotopes can be coupled with ligands that are specific to cell membrane receptors. Thus, WBA and MARG provide data on the distribution and activity of cellular receptors, making them exceptional techniques in the study of cell biology and cell signaling mechanisms (Manuel et al., 2015). In microbiology, the MARG method has been combined with fluorescence in situ hybridization (FISH), using specific oligonucleotides (DNA probes) to identify organisms (Figure 5) (Nielsen & Nielsen, 2005).
WBA can be quantified (quantitative WBA) using dosimetry techniques, i.e., comparing the different radioactivity intensities with radioactive standards of known concentrations. An alternative method to QWBA is autoradioluminography, in which whole-body sections are exposed to a phosphor imaging plate, and scanned by a phosphor image scanner (Figure 4) (Solon & Kraus, 2001)
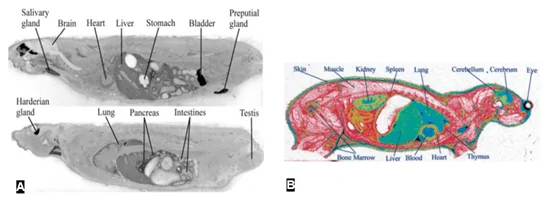
Figure 4 WBA (A) and QWBA (B) of drug distribution in rodents. A – intravenous administration of [14C]-ethanol (Gifford, Espaillat, & Gatley, 2008)darker areas correspond to areas of high ethanol concentration. B – intravenous injection of [3H]-glucose (Potchoiba & Nocerini, 2004). The region of high glucose (radioactivity) concentration is shown in green/blue.
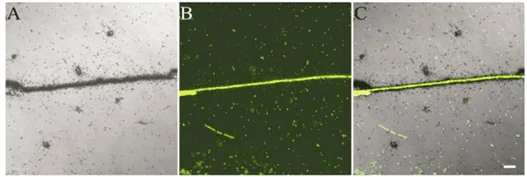
Figure 5: MARG combined with FISH. A–C – the same microscopic view of an activated sludge sample. A– MARG image after incubation of the sample with [14C]-propionate. B The same microscopic field as A, but recorded for fluorescence after hybridization with a probe specific for Meganema perioderoedes (Nielsen & Nielsen, 2005).
Positron-emission tomography (PET) – Diagnostic imaging method used to observe biological processes in the body. In a PET scan, a radioisotope is coupled to a radioactive tracer and then introduced into the body. The radioactive decay from the radiotracer produces positrons: particles with the same mass as electrons, but with opposite charges (i.e., +1). When a positron and an electron collide, they annihilate each other, and two gamma rays are produced, which are detected by a gamma camera. Common radionuclides used in PET scans are listed in Table 1. Gamma cameras record several 2D images that correspond to the radioactivity in the capillary vessels near the target organ or in the whole body. Using multiple projections of the acquired images, a computed tomogram, i.e, a 3D reconstruction, is generated. The 3D tomogram can then be digitally manipulated to analyze the radioactivity distribution in the sample (Figure 6).
Proteins, lipids, sugars, and even patient cells can be used as radioactive tracers. For example, to determine the source of internal bleeding in a patient, it is possible to radiolabel the patient’s red blood cells, inject them back into the body and then follow the accumulation of radioactivity in a specific part of the body using a PET scan (NIH, 2016).
Table 1 Common positron-emitting radioisotopes used in PET
Positron Isotope
| Half-life (min)
| Application
|
11-carbon (11C)
| 20.4
| Detection of tumor margins and metastasis
|
13-nitrogen (13N)
| 9.96
| Myocardial perfusion imaging
|
15-oxygen (15O)
| 2.07
| Hemodynamics and blood flow
|
18-fluor (18F)
| 109.7
| Tumor and metastasis detection
|
68-gallium (68Ga)
| 68.3
| Detection of neuroendocrine tumors
|
(Almuhaideb, Papathanasiou, & Bomanji, 2011; Nakazato, Berman, Alexanderson, & Slomka, 2013; Persoon et al., 2012; Picchio & Castellucci, 2012; Sharma, Singh, Bal, & Kumar, 2014)
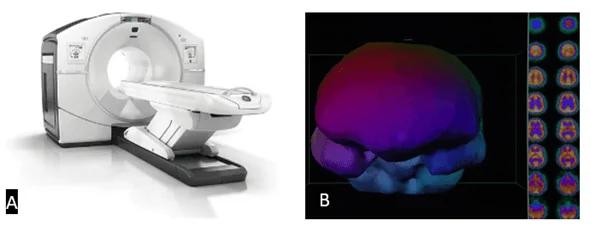
Figure 6 PET / SPECT scan. A – PET scan machine (www.itonline.com). B – Left side: 3D reconstruction (tomogram) of a brain; Right side: 2D sectional images obtained after computed tomography (HBOT, 2019)
Single-photon emission computerized tomography (SPECT) – Similar to PET scan but uses different radiotracers. While in PET scans image comes from the production of positrons, in SPECT imaging, the radioactive tracers emit gamma rays. A list of the most common SPECT radioisotopes is shown in Table 2.
Table 2 Common gamma-ray emitting radioisotopes used in SPECT
Radioisotope
| Half-life
| Application
|
67-gallium (67Ga)
| 78 hours
| Bone disease
|
99-technetium (99mTc)
| 6 hours
| Bone scan Myocardial perfusion scan Brain scan
|
111-indium (111In)
| 2.81 days
| White cell scan
|
123-iodine (123I)
| 13 hours
| Neuroendocrine or neurological tumor scan
|
131-iodine (131I)
| 8.06 days
| Neuroendocrine or neurological tumor scan
|
(Wikipedia, 2019; Wong & Uribe, 2001)
The use of autoradiographic methods brought significant advances to preclinical research. Classic autoradiographic methods are technically straightforward and do not require much expertise from the operators, Moreover, the ability to couple radioisotopes with radiotracers that target radioactive decay to specific tissues made autoradiographic methods highly efficient. However, a major limitation of these techniques is the lack of standard methods to evaluate the binding specificity of radiotracers to their targets. Another limitation of autoradiographic methods is that detection is dependent on the radioactive decay of the radioisotopes used. In the case of long-lived radioisotopes, image development may require several hours of exposure, making it very time-consuming.
In the clinical setting, the controlled used of autoradiography by humans allowed for astonishing advances in human health. However, it should not be overlooked the fact that all the autoradiography methods detailed above make use of ionizing radiation to produce the final images – autoradiographs. In PET and SPECT scans, the radiation to which patients are exposed is higher than in routine chest X-rays exams, due to the internal exposure to the radioactive source. As such, the prescription of PET and SPECT scans must consider health consequences, like increased cancer and neurodegenerative disease risks.
In behavioral neuroscience, the Open Field Test (OFT) remains one of the most widely used assays to evaluate rodent models of affect, cognition, and motivation. It provides a non-invasive framework for examining how animals respond to novelty, stress, and pharmacological or environmental manipulations. Among the test’s core metrics, the percentage of time spent in the center zone offers a uniquely normalized and sensitive measure of an animal’s emotional reactivity and willingness to engage with a potentially risky environment.
This metric is calculated as the proportion of time spent in the central area of the arena—typically the inner 25%—relative to the entire session duration. By normalizing this value, researchers gain a behaviorally informative variable that is resilient to fluctuations in session length or overall movement levels. This makes it especially valuable in comparative analyses, longitudinal monitoring, and cross-model validation.
Unlike raw center duration, which can be affected by trial design inconsistencies, the percentage-based measure enables clearer comparisons across animals, treatments, and conditions. It plays a key role in identifying trait anxiety, avoidance behavior, risk-taking tendencies, and environmental adaptation, making it indispensable in both basic and translational research contexts.
Whereas simple center duration provides absolute time, the percentage-based metric introduces greater interpretability and reproducibility, especially when comparing different animal models, treatment conditions, or experimental setups. It is particularly effective for quantifying avoidance behaviors, risk assessment strategies, and trait anxiety profiles in both acute and longitudinal designs.
This metric reflects the relative amount of time an animal chooses to spend in the open, exposed portion of the arena—typically defined as the inner 25% of a square or circular enclosure. Because rodents innately prefer the periphery (thigmotaxis), time in the center is inversely associated with anxiety-like behavior. As such, this percentage is considered a sensitive, normalized index of:
Critically, because this metric is normalized by session duration, it accommodates variability in activity levels or testing conditions. This makes it especially suitable for comparing across individuals, treatment groups, or timepoints in longitudinal studies.
A high percentage of center time indicates reduced anxiety, increased novelty-seeking, or pharmacological modulation (e.g., anxiolysis). Conversely, a low percentage suggests emotional inhibition, behavioral avoidance, or contextual hypervigilance. reduced anxiety, increased novelty-seeking, or pharmacological modulation (e.g., anxiolysis). Conversely, a low percentage suggests emotional inhibition, behavioral avoidance, or contextual hypervigilance.
The percentage of center time is one of the most direct, unconditioned readouts of anxiety-like behavior in rodents. It is frequently reduced in models of PTSD, chronic stress, or early-life adversity, where animals exhibit persistent avoidance of the center due to heightened emotional reactivity. This metric can also distinguish between acute anxiety responses and enduring trait anxiety, especially in longitudinal or developmental studies. Its normalized nature makes it ideal for comparing across cohorts with variable locomotor profiles, helping researchers detect true affective changes rather than activity-based confounds.
Rodents that spend more time in the center zone typically exhibit broader and more flexible exploration strategies. This behavior reflects not only reduced anxiety but also cognitive engagement and environmental curiosity. High center percentage is associated with robust spatial learning, attentional scanning, and memory encoding functions, supported by coordinated activation in the prefrontal cortex, hippocampus, and basal forebrain. In contrast, reduced center engagement may signal spatial rigidity, attentional narrowing, or cognitive withdrawal, particularly in models of neurodegeneration or aging.
The open field test remains one of the most widely accepted platforms for testing anxiolytic and psychotropic drugs. The percentage of center time reliably increases following administration of anxiolytic agents such as benzodiazepines, SSRIs, and GABA-A receptor agonists. This metric serves as a sensitive and reproducible endpoint in preclinical dose-finding studies, mechanistic pharmacology, and compound screening pipelines. It also aids in differentiating true anxiolytic effects from sedation or motor suppression by integrating with other behavioral parameters like distance traveled and entry count (Prut & Belzung, 2003).
Sex-based differences in emotional regulation often manifest in open field behavior, with female rodents generally exhibiting higher variability in center zone metrics due to hormonal cycling. For example, estrogen has been shown to facilitate exploratory behavior and increase center occupancy, while progesterone and stress-induced corticosterone often reduce it. Studies involving gonadectomy, hormone replacement, or sex-specific genetic knockouts use this metric to quantify the impact of endocrine factors on anxiety and exploratory behavior. As such, it remains a vital tool for dissecting sex-dependent neurobehavioral dynamics.
The percentage of center time is one of the most direct, unconditioned readouts of anxiety-like behavior in rodents. It is frequently reduced in models of PTSD, chronic stress, or early-life adversity. Because it is normalized, this metric is especially helpful for distinguishing between genuine avoidance and low general activity.
Environmental Control: Uniformity in environmental conditions is essential. Lighting should be evenly diffused to avoid shadow bias, and noise should be minimized to prevent stress-induced variability. The arena must be cleaned between trials using odor-neutral solutions to eliminate scent trails or pheromone cues that may affect zone preference. Any variation in these conditions can introduce systematic bias in center zone behavior. Use consistent definitions of the center zone (commonly 25% of total area) to allow valid comparisons. Software-based segmentation enhances spatial precision.
Evaluating how center time evolves across the duration of a session—divided into early, middle, and late thirds—provides insight into behavioral transitions and adaptive responses. Animals may begin by avoiding the center, only to gradually increase center time as they habituate to the environment. Conversely, persistently low center time across the session can signal prolonged anxiety, fear generalization, or a trait-like avoidance phenotype.
To validate the significance of center time percentage, it should be examined alongside results from other anxiety-related tests such as the Elevated Plus Maze, Light-Dark Box, or Novelty Suppressed Feeding. Concordance across paradigms supports the reliability of center time as a trait marker, while discordance may indicate task-specific reactivity or behavioral dissociation.
When paired with high-resolution scoring of behavioral events such as rearing, grooming, defecation, or immobility, center time offers a richer view of the animal’s internal state. For example, an animal that spends substantial time in the center while grooming may be coping with mild stress, while another that remains immobile in the periphery may be experiencing more severe anxiety. Microstructure analysis aids in decoding the complexity behind spatial behavior.
Animals naturally vary in their exploratory style. By analyzing percentage of center time across subjects, researchers can identify behavioral subgroups—such as consistently bold individuals who frequently explore the center versus cautious animals that remain along the periphery. These classifications can be used to examine predictors of drug response, resilience to stress, or vulnerability to neuropsychiatric disorders.
In studies with large cohorts or multiple behavioral variables, machine learning techniques such as hierarchical clustering or principal component analysis can incorporate center time percentage to discover novel phenotypic groupings. These data-driven approaches help uncover latent dimensions of behavior that may not be visible through univariate analyses alone.
Total locomotion helps contextualize center time. Low percentage values in animals with minimal movement may reflect sedation or fatigue, while similar values in high-mobility subjects suggest deliberate avoidance. This metric helps distinguish emotional versus motor causes of low center engagement.
This measure indicates how often the animal initiates exploration of the center zone. When combined with percentage of time, it differentiates between frequent but brief visits (indicative of anxiety or impulsivity) versus fewer but sustained center engagements (suggesting comfort and behavioral confidence).
The delay before the first center entry reflects initial threat appraisal. Longer latencies may be associated with heightened fear or low motivation, while shorter latencies are typically linked to exploratory drive or low anxiety.
Time spent hugging the walls offers a spatial counterbalance to center metrics. High thigmotaxis and low center time jointly support an interpretation of strong avoidance behavior. This inverse relationship helps triangulate affective and motivational states.
By expressing center zone activity as a proportion of total trial time, researchers gain a metric that is resistant to session variability and more readily comparable across time, treatment, and model conditions. This normalized measure enhances reproducibility and statistical power, particularly in multi-cohort or cross-laboratory designs.
For experimental designs aimed at assessing anxiety, exploratory strategy, or affective state, the percentage of time spent in the center offers one of the most robust and interpretable measures available in the Open Field Test.
Written by researchers, for researchers — powered by Conduct Science.








Monday – Friday
9 AM – 5 PM EST
DISCLAIMER: ConductScience and affiliate products are NOT designed for human consumption, testing, or clinical utilization. They are designed for pre-clinical utilization only. Customers purchasing apparatus for the purposes of scientific research or veterinary care affirm adherence to applicable regulatory bodies for the country in which their research or care is conducted.