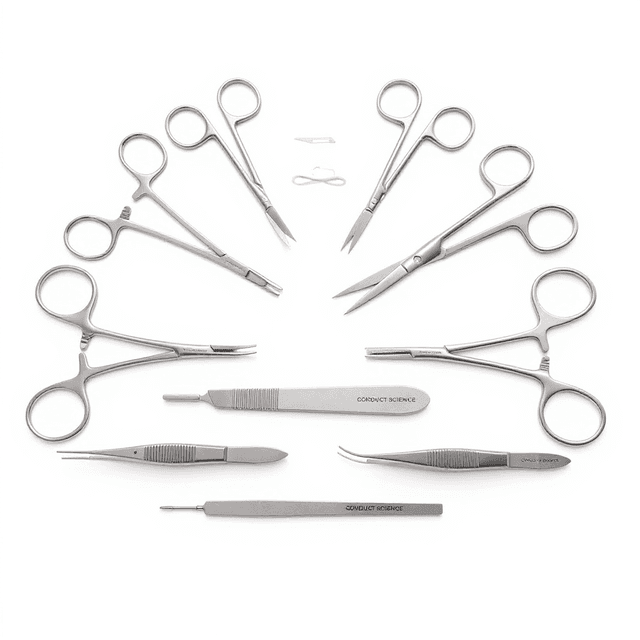
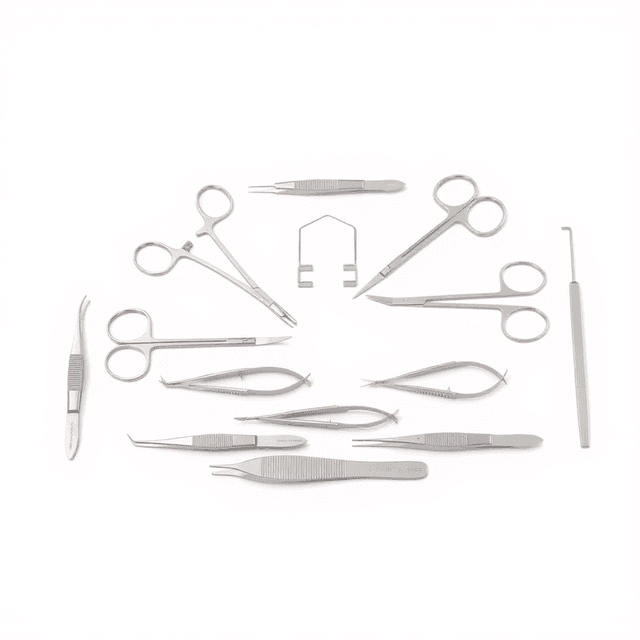
Cardiovascular Surgery Instrument Kit
Cardiovascular surgery kits focus on the heart and blood vessels. Cardiac surgery kit is mainly used to treat the complexities of ischemic heart diseases, congenital heart diseases, valvular heart diseases, endocarditis, rheumatic heart diseases, and atherosclerosis.

Louise Corscadden, PhD
Director of Science · ConductScience
Ask Louise about Cardiovascular Surgery Instrument Kit fit, setup, configuration, or quote prep.
Already working with us? Sign in to connect this with My Scientist.
Mouse Kit
| Operating Scissors (Round Type) | S/S Str/11.5cm |
| IRIS-Fine Scissors (Round Type) | S/S Str/9.5cm |
| Spring Scissors (Triangular) | S/S Str/5*0.1mm/8.5cm |
| IRIS Dissecting Forceps-Large | Cvd, 0.8mm Tips, 10cm |
| Dressing Forceps | Str, 1.8mm Tips, 9cm |
| HARTMAN Mosquito Forceps | Str, 1.0mm Tips, 10cm |
| HARTMAN Mosquito Forceps | Cvd, 1.0mm Tips, 10cm |
| PGA Sutures w/Needle | o1/2/4×10/90cm/5-0 (50/Box |
| Sutures w/Needle | △3/8/2.5×7/30cm/6-0 (50/Box) |
| STEVENS Hooks, 1 Angled Tooth | (5mm long), 12.5cm |
| 3×3 Teeth Retractors-Blunt | 4.5cm |
| OLSEN-HEGAR Needle Holders with Scissors | Str, 12cm |
| SS Micro Clamps | Str/L*W 4*0.75mm/13mm |
| Clip Applicator for R31005- and R31006-Clamps | 14cm |
| Spinal Cord Hook | Tip Dia. 3mm/12cm |
| Instrument Storage Portfolio | 32*22cm |
Rat Kit
| Operating Scissors (Round Type) | S/S Str/12.5cm |
| IRIS-Fine Scissors (Round Type) | S/S Str/10.5cm |
| Spring Scissors (Triangular) | S/S Str/5*0.1mm/8.5cm |
| IRIS Dissecting Forceps-Large | Cvd, 0.8mm Tips, 10cm |
| IRIS Dissecting Forceps | Str, 0.8mm Tips, 10cm |
| HALSTED Mosquito Forceps | Str, 1.0mm Tips, 12.5cm |
| HALSTED Mosquito Forceps | Cvd, 1.0mm Tips, 12.5cm |
| PGA Sutures w/Needle | o1/2/4×10/90cm/5-0 (50/Box) |
| Sutures w/Needle | △3/8/2.5×7/30cm/6-0 (50/Box) |
| STEVENS Hooks, 1 Angled Tooth (5mm long) | 12.5cm |
| 3×3 Teeth Retractors-Blunt | 4.5cm |
| OLSEN-HEGAR Needle Holders with Scissors | Str, 12cm |
| SS Micro Clamps | Str/L*W 4*0.75mm/13mm |
| Spinal Cord Hook | Tip Dia. 3mm/12cm |
| Clip Applicator for R31005- and R31006-Clamps | 14cm |
| Instrument Storage Portfolio | 32*22cm |
Introduction
Cardiovascular surgery is mainly performed to treat the complexities of ischemic heart diseases, congenital heart diseases, valvular heart diseases, endocarditis, rheumatic heart diseases, and atherosclerosis. It also includes heart transplantation.
The preferred rodent species for conducting cardiovascular research is shifting from rat to mouse. The main advantage of using the rat is its bigger size. However, the development of more sophisticated micro-dissecting microscopes and imaging devices, high-caliber microsurgical instruments, small catheters for hemodynamic measurements, etc., has made microsurgery in the mouse more feasible than rats. Mouse models mimicking human diseases are essential tools in biomedical research, which aim to understand the underlying mechanisms of many disease states. Mouse models have gained popularity because of their small size, rapid gestation age (21 days), relatively low husbandry costs, and convenience in housing and handling. Moreover, the extensively characterized mouse genome and gene-targeting (i.e., knockout) and transgenic overexpression experiments are widely performed using mice rather rats (Tarnavski, 2009).
Pre-operative Set-up and Anesthesia Induction
Thoroughly clean and sterilize the instruments used for the surgery. Autoclave and disinfect the equipment. Clear the operating area of any disturbances and ensure asepsis. Before the surgery, record the subject identification details such as strain and gender, most importantly note the weight of the subject. Also, examine the subject physically to assess its health status and activeness. Ensure that the subject has been acclimated to the facility appropriately. The acclimation process can last from a few days to a couple of weeks.
Anesthesia
- Anesthetize the surgical subjects with pentobarbital 70 mg/kg in a solution of 15 mg/ml (Use a face mask or an anesthetic chamber to anesthetize the animals). Do not administer analgesics pre-operatively.
- Shave the chest with a hair clipper or a razor.
- Place the animal on the surgical stage in surgical position for the subsequent intubation, wire 3-0 silk behind the front incisors, pull taut and fix with tape.
- Intubate the animal with a 20-ga catheter, attached to the extender.
- Connect the intubation tube to the ventilator and start ventilating the animal.
- Perform the surgical procedure at room temperature (except for the ischemia-reperfusion).
- Prepare the surgical field.
Cardiovascular Surgery Protocols
Mouse Cardiac Surgery Protocol (Tarnavski et al., 2004)
- Open the chest with a lateral incision at the 4th intercostal space on the left side of the sternum and retract the chest.
- Remove the pericardial sac and pass an 8-0 silk suture in the myocardium at the anterior surface of the heart for one second and then remove it.
- Close the chest wall by approximating the third and fourth ribs with one or two interrupted sutures, return the muscles to their original position, and the skin closed with retract4-0 prolene suture.
- Gently disconnect the animal from the ventilator.
Aortic Banding (Pressure-overload Model) Method (Tarnavski, 2009)
- Locally anesthetize the animal by injecting 0.1 ml of 0.2% lidocaine subcutaneously at the surgical site.
- Transversely incise the skin 5-mm with scissors 1–2 mm higher than the level of the ‘‘armpit’’ (with paw extended at 90o) 2 mm away from the left sternal border.
- Separate the two layers of thoracic muscles.
- Separate the intercostal muscles in the 2d intercostal space.
- Interpolate the chest retractor to facilitate the view.
- Pull the thymus and surrounding fat behind the left arm of the retractor. Gently pull the pericardial sac and attach it to both arms of the retractor.
The steps mentioned above remain the same for both ascending aortic constriction and transverse aortic constriction.
Ascending Aortic Constriction (AAC)
- Bluntly dissect the ascending portion of the aorta on its lateral side from the pulmonary trunk with Foerster curved forceps.
- Place Foerster curved forceps from the medial side under the ascending aorta, hold the 7-0 silk suture on the opposite side and pass it underneath the aorta.
- Tie a loose double knot to create a loop 7–10mm in diameter.
- Position a needle of proper size into the circuit.
- Tie the loop around the aorta and needle, and bind with the second knot; immediately remove the needle to provide a lumen with a stenotic aorta. Make another knot to secure the tie.
- Remove the chest retractor and re-inflate the lungs.
- Close the chest wound layer-by-layer.
Transverse Aortic Constriction (TAC)
- Bluntly depart the thymus, and pericardial sac with slightly curved forceps, then separate the aortic arch from the surrounding tissues and vessels.
- With the help of a blunted needle, create a way underneath the aortic arch.
- With a ‘‘wire and snare’’ device, deliver a 7-0 silk suture underneath the aortic arch between the innominate and left carotid arteries.
- Tie a loose double knot to create a loop 7–10mm in diameter.
- Position a needle of proper size into the circuit.
- Tie the circle around the aorta and needle, and secure with the second knot; remove the needle immediately to provide a lumen with a stenotic aorta. Make another knot to bind the tie.
- Remove the chest retractor and re-inflate the lungs.
Pulmonary Artery Banding (Right Ventricular Pressure-overload Model) Method
The principle of pulmonary artery banding is similar to that of aortic banding. However, while performing this surgery, be careful of specific challenges.
- Approach the great vessels via the second intercostal space.
- To supplement the animal with local anesthesia inject 0.1 ml of 0.2% lidocaine subcutaneously at the surgical site.
- Make a transverse 5-mm incision of the skin with scissors 1–2 mm higher than the level of the ‘‘armpit’’ (with paw extended at 90o) 2 mm away from the left sternal border.
- Separate the two layers of thoracic muscle.
- Isolate the intercostal muscles in the 2d intercostal space.
- Interpolate the chest retractor to visualize the organs.
- Pull the thymus and surrounding fat behind the left arm of the retractor. Gently draw the pericardial sac and attach it to both the arms of the retractor.
- After moving the pericardium, observe the pulmonary trunk (partially covered by the left atrium).
- With the help of Foerster curved forceps, bluntly dissect the pulmonary trunk from the aorta (on the left) and left atrium (on the right).
- With a blunted needle, create a passage under the pulmonary trunk.
- With the help of a ‘‘wire and snare’’ device, move the 7-0 silk suture underneath the pulmonary trunk.
- Tie a loose double knot to create a loop 7–10mm in diameter.
- Position an appropriately sized needle into the loop.
- Tie the loop around the aorta and needle and secure with the second knot; immediately remove the needle to provide a lumen with a stenotic pulmonary artery. Make another knot to secure the tie.
- As the heart rate significantly slows down, stimulate the mouse.
- Remove the chest retractor and re-inflate the lungs.
- Close the chest wound layer-by-layer.
Myocardial Infarction Model (Permanent Ligation of the Artery) Protocol
- For local anesthesia, inject 0.1 ml of 0.2% lidocaine subcutaneously at the surgical site.
- Make an oblique 8-mm incision of the skin (parallel to ribs) 2 mm far from the left sternal border toward the left armpit (1–2 mm below it).
- Separate the two layers of the thoracic muscle.
- Separate the intercostal muscles in the fourth intercostal space.
- Insert the chest retractor to facilitate the view.
- Gently pull apart the pericardial sac and attach it to both arms of the retractor.
- Locate the left anterior descending (LAD) coronary artery.
- Pass the tapered needle with 7-0 silk suture underneath the LAD.
- Tie the suture with one double and then with one single knot.
- Remove the chest retractor and close the wound layer-by-layer.
Ischemia-Reperfusion Model Protocol
- Hold the artery and place the suture (same as step 1-8 above).
- To create ischemia, tie the temporary suture around the LAD. Cover the incision with wet gauze to prevent drying.
- Release the tie (do not remove the suture) at the end of ischemia and observe the reperfusion.
- If the procedure is survival, leave the suture underneath the LAD and close the chest wound layer-by-layer.
- Retie the suture to demarcate the area at risk by infusion of blue dye.
Coronary Artery Ligation and Intra-myocardial Injection in a Murine Model of Infarction Protocol (Virag & Lust, 2011)
- Intubate the animals intratracheally by transesophageal illumination using a fiber-optic light.
- Connect the tubing to the ventilator.
- Expose the pectoral muscles by making an incision on the chest to view the ribs.
- For ischemia/reperfusion studies, place a 1 cm piece of PE tubing over the heart to tie the ligature to so that occlusion/reperfusion can be customized.
- For intramyocardial injections, use a syringe with a sterile 30gauge beveled needle.
- Complete the myocardial manipulations, close the rib cage, the pectoral muscles, and the skin sequentially.
- Apply 0.25% Bupivacaine in sterile saline to muscle layer before the closure of the skin.
- Give a subcutaneous injection of saline and place the mice in a warming chamber until they are sternally recumbent.
- Return the animals to the vivarium and housed under standard conditions until the time of tissue collection.
- Anesthetize the mice, arrest the heart in diastole with KCl or BDM, rinse with saline, and immerse in fixative.
- Subsequently, perform routine procedures for processing, embedding, sectioning, and histological staining.
Postoperative Management
- Administer the first dose of the analgesic (0.1 mg/kg) intraperitoneally at the completion of the surgery.
- Move the animal to another ventilator in the recovery area with 100% oxygen loosely connected to its inflow.
- Provide heat by either a 60-W lamp or a heating pad at a low setting.
- Once the mouse attempts to breathe spontaneously (generally after 45–60 min), disconnect the intubation tube from the ventilator.
- Keep the intubation tube in the trachea for another 10–15 min until the mouse resumes the regular breathing pattern.
- Provide supplementary oxygen (the mouse is placed next to the source of oxygen).
- Extubate the mouse and return it to a clean, warm cage.
Applications
Genomic Study of Coronary Diseases in Murine Models
Mouse models mimicking human cardiac diseases are essential paradigms to explain the underlying mechanisms of many disease states. Several surgical models have been developed that mimic human myocardial infarction (MI) and pressure-overload-induced cardiac hypertrophy. Microarray technologies, used in the research, measure the expression of thousands of genes simultaneously, allowing for the identification of genes and pathways that may potentially be involved in the disease process. In the study, a description of three major surgical procedures has been discussed: 1) aortic constriction, 2) pulmonary artery banding, 3) MI (including ischemia-reperfusion). The cardiac surgery techniques mentioned have been and will continue to be, essential for elucidating the molecular mechanisms of cardiac hypertrophy and genome profiling.
Evaluation of Galectin-1: a Biomarker of Surgical Stress in Murine Model of Cardiac Surgery (Hashmi & Al-Salam, 2015)
Galectin-1 (GAL-1) is a member of the β-galactoside-binding lectins family. GAL-1 regulates cell-cell and cell-matrix interactions, the immune response, apoptosis, cell cycle, RNA splicing, and neoplastic transformation. Satwat and Suhail investigated the effect of heart manipulation secondary to cardiac surgery on the level of GAL-1 in murine heart and plasma. They used male C57B6/J mice for the adopted model of cardiac surgery. Then the heart samples were processed for immunohistochemical and immunofluorescent labeling; Enzyme-linked immunosorbent assay and quantitative RT-PCR to identify GAL-1 levels in the heart and plasma during the first 24 hours following cardiac surgery. Results suggested that the GAL-1 is a valuable biomarker of surgical stress.
Evaluating Coronary Artery Ligation and Intramyocardial Injection in a Murine Model of Infarction
Mouse models are widely used to study acute injury and chronic remodeling of the myocardium in vivo. With the advancement of genetic modifications to the whole organism or the myocardium and an array of biological and synthetic materials, there is an excellent potential for any combination of these to mitigate the extent of the acute ischemic injury and impede the onset of heart failure according to myocardial remodeling.
The study presented the methods and materials used to perform the microsurgery reliably and the modifications for temporary (with reperfusion) or permanent coronary artery occlusion studies as well as intramyocardial injections. Development of miniature technology for imaging in vivo, analyzing large-scale genomics and proteomics, drug screening, efficacy of cell-based and/or protein therapies as well as biomaterials, combined with the increasingly wide range of genetic manipulations afforded by ubiquitous or tissue-specific transgenic or mutant/knockout mice, the murine model of myocardial infarction, undoubtedly, continue to be an invaluable tool in evaluating acute cardiac injury and long-term remodeling. Therefore, there is an absolute value in being able to perform these experiments reliably and reproducibly.
Precautions
Murine cardiac surgical procedures are relatively short, so it is not necessary to withhold food and water from mice before the surgery. Before starting the surgical procedure, ensure the adequate depth of the anesthesia using a toe pinch test or other anesthesia assessment tests. It is important not to disturb or agitate the animal while the anesthetic is taking effect to ensure smooth induction and facilitate subsequent procedures. Do not administer pre-surgical analgesia (buprenorphine) since narcotic analgesics are known to depress the respiratory center and may interfere with the survival after the open-chest surgery. Since the described surgical procedures are relatively short (15–20 min), it is not critical to strictly regulate the core temperature of the mouse. Do not pull apart the pericardial sac with too much force as it may rupture the wall of the left superior vena cava and cause bleeding that can be fatal. The significant challenges that arise during the cardiac surgery are because of the extremely thin and delicate walls of the pulmonary trunk and the incapacity of the right ventricle to undergo stress while the pulmonary artery is being manipulated. Carry out the whole surgical procedure under aseptic conditions.
Strengths and Limitations of Rodent Models for Cardiac Surgery
The most widely used rodent species for conducting cardiovascular research is the mouse. The significant advantage of using the rat is its bigger size. However, the development of micro-dissecting microscopes and other imaging devices, high-caliber microsurgical instruments, small catheters for hemodynamic measurements, etc., has made microsurgery in the mouse as feasible as in the rat. Mouse models have gained popularity because of their small size, rapid gestation age (21 days), relatively low maintenance costs, convenience in housing and handling, and fewer compound requirements for pharmacological studies. Use of a simple dissecting scope or magnifying glass and well-lit conditions enable the vasculature to be seen readily. To exterminate the risk of postoperative mortality, it is essential to avoid severing large vessels since the total blood volume of a 25g mouse is less than 2ml. If excessive bleeding occurs, gentle application of pressure or pinpoint cauterization can be used to stop the bleeding. Also, the mouse genome has been extensively characterized, and gene-targeted (i.e., knockout) and transgenic overexpression experiments are more commonly performed using mice rather than rats.
Summary
- Cardiovascular surgery focuses on the heart and blood vessels. Cardiac surgery is mainly performed to treat the complexities of ischemic heart diseases, congenital heart diseases, valvular heart diseases, endocarditis, rheumatic heart diseases, and atherosclerosis.
- Mouse models have gained popularity because of their small size, rapid gestation age (21 days), relatively low husbandry costs, convenience in housing and handling, and fewer compound requirements for pharmacological studies.
- With the advancement of genetic modifications to the whole organism or the myocardium and an array of biological and synthetic materials, there is an excellent potential for any combination of these to mitigate the extent of the acute ischemic injury and impede the onset of heart failure pursuant to myocardial remodeling.
- Do not administer pre-surgical analgesia (buprenorphine) since narcotic analgesics are known to depress the respiratory center and may interfere with the survival after the open-chest surgery.
- The significant challenges that arise during cardiac surgery are the extremely thin and delicate walls of the pulmonary trunk and the incapacity of the right ventricle to undergo stress while the pulmonary artery is being manipulated. Be careful during the operative procedures.
References
- Hashmi, S., & Al-Salam, S. (2015). Galectin-1: a biomarker of surgical stress in a murine model of cardiac surgery. Int J Clin Exp Pathol, 8, 7157-7164.Tarnavski, O. (2009). Mouse surgical models in cardiovascular research. Methods Mol Biol, 573, 115-37.Tarnavski, O., McMullen, J. R., Schinke, M., Nie, Q., Kong, S., & Izumo, S. (2004). Mouse cardiac surgery: comprehensive techniques for the generation of mouse models of human diseases and their application for genomic studies. Physiol Genomics, 16, 349-360.Virag, J. A., & Lust, R. M. (2011). Coronary Artery Ligation and Intramyocardial Injection in a Murine Model of Infarction. J Vis Exp, 52, 2581.
How It Works
Cardiovascular microsurgery in small animal models requires instruments scaled to rodent anatomy with sufficient precision for vessel manipulation and tissue handling. The operating scissors provide initial access through skin and muscle layers, while iris fine scissors enable precise dissection around delicate cardiac structures. Spring scissors with triangular tips allow fine trimming of suture material and small vessel branches.
Forceps selection is critical for tissue handling without damage. The graduated tip sizes (0.8mm iris forceps for delicate vessel handling, 1.8mm dressing forceps for general tissue manipulation) provide appropriate grip strength for different tissue types. Hartman mosquito forceps in straight and curved configurations enable hemostasis control during the procedure.
The micro clamps (4×0.75mm dimensions) provide temporary vascular occlusion for anastomosis procedures, while the needle holder with integrated scissors streamlines suturing workflow. The spinal cord hook facilitates tissue retraction and organ positioning during complex cardiac access procedures.
Features & Benefits
scissors_lengths
- 8.5cm to 12.5cm
forceps_tip_sizes
- 0.8mm to 1.8mm
forceps_lengths
- 9cm to 12.5cm
suture_sizes
- 5-0 and 6-0
suture_needle_types
- ○1/2/4×10 and △3/8/2.5×7
suture_lengths
- 30cm to 90cm
sutures_per_box
- 50
micro_clamp_dimensions
- L×W 4×0.75mm
hook_tip_diameter
- 3mm
retractor_teeth
- 3×3
storage_portfolio_size
- 32×22cm
anesthesia_dosage
- pentobarbital 70 mg/kg in 15 mg/ml solution
intubation_catheter
- 20-ga
suture_wire
- 3-0 silk
Automation Level
- manual
Species
- Mouse
- Rat
Material
- S/S (Stainless Steel)
Brand
- RWD
Research Domain
- Aging Research
- Cardiovascular
- Metabolic Research
- Neuroscience
- Toxicology
Weight
- 4.41 kg
Dimensions
- L: 34.0 mm
- W: 39.0 mm
- H: 33.0 mm
| Feature | This Product | Typical Alternative | Advantage |
|---|---|---|---|
| Instrument Length Range | 8.5cm to 12.5cm species-optimized lengths | Generic lengths not optimized for specific rodent anatomy | Provides appropriate leverage and access angles for each target species anatomical scale. |
| Forceps Tip Graduation | Three distinct tip sizes (0.8mm, 1.0mm, 1.8mm) | Limited tip size options in basic surgical sets | Enables appropriate grip strength selection from delicate vessel handling to general tissue manipulation. |
| Suture Variety | Dual gauge (5-0, 6-0) with multiple needle configurations | Single suture type requiring separate purchasing | Provides complete suture options for different tissue layers and closure requirements in one kit. |
| Specialized Cardiovascular Tools | Includes micro clamps (4×0.75mm) and cardiac hooks | General surgical kits lack cardiovascular-specific instruments | Eliminates need for separate procurement of specialized vascular occlusion and retraction tools. |
| Organization System | Dedicated 32×22cm instrument portfolio | Basic instrument trays or no organization system | Maintains sterile presentation and systematic instrument arrangement throughout procedures. |
This kit provides species-optimized instrument sizing with graduated forceps capabilities and comprehensive suture options specifically configured for cardiovascular research applications. The inclusion of specialized micro clamps and cardiovascular-specific tools distinguishes it from general surgical instrument sets.
| Model | SKU | Listed price | Status | Dimensions |
|---|---|---|---|---|
| Rat | RWD-SP0008-R | $889.00 | Available | 34.0 x 39.0 x 33.0 cm |
| Mouse | RWD-SP0008-M | $849.00 | Available | 34.0 x 39.0 x 33.0 cm |
Practical Tips
Inspect cutting instrument alignment before each procedure by observing blade contact under magnification.
Why: Misaligned cutting edges reduce precision and can cause tissue trauma during dissection.
Arrange instruments in order of use sequence before beginning procedures to minimize sterile field disruption.
Why: Systematic organization reduces procedure time and maintains sterile technique throughout surgery.
Verify micro clamp occlusion pressure by testing on similar diameter tubing before vessel application.
Why: Appropriate clamp pressure ensures hemostasis without vessel wall damage that could compromise anastomosis.
Use spring scissors for suture cutting rather than larger instruments to prevent inadvertent tissue damage.
Why: The controlled cutting action and fine tips reduce risk of cutting adjacent tissues during suture trimming.
If forceps lose grip strength, check for debris accumulation in spring mechanisms and clean thoroughly.
Why: Accumulated tissue debris reduces spring tension and compromises precise tissue handling capability.
Document specific instrument configurations used for each procedure to ensure reproducible surgical techniques.
Why: Consistent instrument selection reduces procedural variability that could affect experimental outcomes.
Select suture gauge based on tissue thickness rather than general procedure type for optimal healing.
Why: Appropriate suture sizing minimizes foreign body reaction while providing adequate tensile strength for tissue healing.
Setup Guide
What’s in the Box
- Operating scissors (round type, species-specific length)
- Iris fine scissors (round type)
- Spring scissors (triangular)
- Iris dissecting forceps (0.8mm tips)
- Dressing forceps (1.8mm tips)
- Hartman mosquito forceps (straight and curved)
- PGA sutures with needles (5-0 gauge, 50/box)
- Sutures with needles (6-0 gauge, 50/box)
- Stevens hooks (1 angled tooth)
- 3×3 teeth retractors
- Olsen-Hegar needle holders with scissors
- Stainless steel micro clamps
- Clip applicator for micro clamps
- Spinal cord hook (3mm tip diameter)
- Instrument storage portfolio (32×22cm)
Warranty
ConductScience provides a standard one-year manufacturer warranty covering defects in materials and workmanship, with technical support for proper instrument care and sterilization procedures.
Compliance
What sterilization methods are compatible with these stainless steel instruments?
Standard autoclave sterilization at 121°C is appropriate for stainless steel construction. Chemical sterilization with glutaraldehyde or ethylene oxide is also compatible, though autoclave sterilization is preferred for routine use.
How do I select between the mouse and rat kit configurations?
Kit selection depends on target species anatomy. Mouse kits feature 11.5cm operating scissors optimized for smaller thoracic cavities, while rat kits include 12.5cm instruments providing appropriate reach for larger anatomical structures.
What is the appropriate suture gauge selection for different cardiovascular procedures?
Use 5-0 PGA sutures for vessel anastomosis and major tissue closure, while 6-0 sutures are appropriate for fine tissue approximation and superficial layer closure. Needle configuration should match tissue penetration requirements.
How should micro clamps be applied to avoid vessel damage?
Apply micro clamps with minimal pressure sufficient for hemostasis. The 4×0.75mm dimensions provide appropriate vessel compression without crushing. Limit application time to prevent tissue ischemia during anastomosis procedures.
What maintenance is required for maintaining instrument sharpness?
Sharp instruments should be inspected before each use and resharpened when cutting performance degrades. Avoid contact with hard surfaces and store in the provided portfolio to prevent edge damage between procedures.
Can these instruments be used for non-cardiovascular procedures?
While optimized for cardiovascular surgery, these instruments are suitable for other microsurgical procedures requiring precise tissue handling in small animal models, including neurological and hepatic surgeries.
How do I determine appropriate anesthesia depth for cardiovascular procedures?
The kit attributes suggest pentobarbital at 70 mg/kg in 15 mg/ml solution, though anesthesia protocols should be determined by veterinary consultation and institutional guidelines for specific procedures.
Have a question about this product?
Accessories
Enhance your setup with compatible accessories